BioByte 062: First RNA-editing trial in US, an integrated classification of Parkinson's, shortening CAR-T manufacturing, cell surface RNAs control neutrophil recruitment
Welcome to Decoding Bio, a writing collective focused on the latest scientific advancements, news, and people building at the intersection of tech x bio. Happy decoding!
Sad that the Lions or Ravens aren’t in the Super Bowl? Unhappy that Travis Kelce has a chance to win the Super Bowl when he’s already won dating? Well luckily for you we have some SCIENCE here to keep you comfort as you cry yourself to sleep over the next few weeks and question everything. Just do us a favor and don’t crush your computer in the embrace of loneliness…
What we read
Blogs
Vertex one step closer to a major blockbuster? [Vertex, January 2024]
The excitement: Analysts and biopharma have been carefully watching the phase III readout of VX-548, touted to have blockbuster potential.
Indication: moderate to severe acute pain, which 80M+ seek treatment for in the USA alone
MoA = Novel NaV1.8 blockade in peripheral nervous system
Rationale: non-addictive (unlike opioids)
Trial results: met primary endpoint but MISSED key secondary endpoint of superiority to the opioid vicodin.
Planned submission to the FDA for approval by mid-2024.
Verdict: although the secondary endpoint was missed, Vertex tout VX-548 is primarily solving for safety and tolerability, rather than efficacy. Time will tell whether the molecule will live up to its lofty expectations.
Spill the TEA: Making economically viable bioproducts [Elliott Hershberg, January 2024]
Synthetic biology has promised a revolution in how we manufacture and produce everyday items from foods to chemicals and clothing. However, such a revolution has been slow to arrive. Ginkgo, one of the industry’s most prominent ventures has lost 90% of its market value since its 2021 IPO.
As well as technical challenges, a major contributing factor is that many synthetic biology businesses are not financially competitive in our current society. In this blog, Hershberg outlines the concept of Techno-Economic Analysis (TEA) for helping to understand a new technology’s ability to compete in an industrial market and where the highest leverage opportunities for improvement lie. He outlines tools for synbio companies to automate this analysis such as Scaler and how this could provide for better project financing through cross-company synergies.
Toward the eradication of medical diagnostic errors [Topol, Science, January 2024]
The medical community faces a significant challenge with diagnostic errors, with studies estimating that nearly 800,000 Americans die or are permanently disabled by diagnostic errors annually. Despite advances in medical technology, the problem persists, driven partly by the limited time available for doctors to evaluate patients, relying on quick and often error-prone System 1 thinking.
AI is emerging as a potential solution to improve diagnostic accuracy. Recent studies demonstrate that AI, especially deep learning models, can enhance the accuracy of diagnoses, reduce workload, and identify elusive conditions. However, concerns about bias and doctors' acceptance of AI's decisions need to be addressed. The future holds promise for AI to serve as a valuable tool, providing second opinions and augmenting medical expertise, ultimately working towards the goal of eradicating diagnostic errors. This commentary in Science offers a brief literature review of some of the exciting advancements in AI diagnostics, as well as the barriers ahead.
First-ever RNA editing trial in US gets clearance, pitting ‘exon editing’ technology against vision loss [Ryan Cross, Endpoints, 2023]
Ascidian Therapeutics has received approval to commence testing its RNA editing therapy. This therapy is the first of its kind to enter clinical trials.
The treatment edits RNA to correct two-thirds of the genetic mutations responsible for Stargardt disease, a type of inherited vision loss. RNA editing's transient nature potentially makes it safer than permanent DNA editing techniques like CRISPR/Cas9. Administered via AAV, Ascidian's exon editing strategy aims to treat the disease, which is associated with over 1000 mutations. These are too many mutations to create individual treatments for, and the gene involved is too large for delivery. By hijacking the RNA splicing process, the company believes it can treat the disease where most other therapies cannot.
Emerging Cell Therapies [Jocelynn Pearl, Jan 2024]
Jocelynn Pearl summarized some key updates from the Keystone Symposium on Emerging Cellular Therapies. A few highlights:
Drew Weissman (recent winner of the Nobel Prize for his work on nucleoside base modifications that enabled mRNA vaccines against COVID-19) stressed the importance of custom mRNA modifications for specific cell types to optimize expression efficiency, rather than a one-size-fits-all-cell-types modification.
Unpublished work from Crystal Chen from Stanley Qi’s lab at Stanford on a screen and reporter system for engineering transcriptional control elements to optimize gene expression in activated T cells.
Positive clinical data for 2 cell therapies for neurological indications:
Neurona’s NRTX-1001, an allogeneic cell therapy derived from human pluripotent stem cells and differentiated into interneurons that secrete the inhibitory neurotransmitter GABA to dampen excessive neuronal firing in epileptic seizures, decreased seizure activity in 3 patients.
BlueRock Therapeutics presented clinical data for their cell therapy for Parkinson’s disease, an allogeneic cell therapy designed to replace the dopamine producing neurons that are lost in a deep brain structure called the substantia nigra in PD.
Academic papers
A biological classification of Parkinson's disease: the SynNeurGe research diagnostic criteria [Günter U Höglinger et al., The Lancet, January 2024]
Why it matters: The SynNeurGe classification system represents a significant advancement in Parkinson's disease (PD) research, shifting the focus from a primarily clinical diagnosis to one that is more biologically based. By integrating biomarkers and genetic data, this system offers a more nuanced understanding of the disease's onset and progression. This approach is crucial for developing targeted, effective therapies and moves the field closer to precision medicine. Understanding the biological underpinnings of PD is essential for the development of disease-modifying treatments and for addressing the diverse manifestations of this complex condition.This paper introduces a new classification system for PD aimed at enhancing research and understanding of the disease. This system, termed SynNeurGe, incorporates three key components: the presence or absence of pathological α-synuclein (S) in tissues or CSF, evidence of neurodegeneration (N) as indicated by neuroimaging, and the identification of specific genetic variants (G) linked to PD. This classification acknowledges the complexity and heterogeneity of PD and is designed to be used solely for research purposes at this stage.
Development of Ingenui-T, a Novel Vein-to-Vein Solution for Rapid Autologous CAR T-Cell Manufacturing Starting From Whole Blood, for the Treatment of Autoimmune Diseases [Anaya et al., BioRxiv, Jan 2024]
Why it matters: CAR-T therapies require an invasive and time consuming medical procedure called apheresis as a first step. Kyverna Therapeutics has developed an alternative approach using whole blood, resulting in a shorter and more efficient cell therapy manufacturing process. Apheresis, a medical procedure that involves removing whole blood from a patient and separating the blood into individual components so that one particular component can be removed, is typically the first step in cell therapy to acquire starting material for cell therapy products. The procedure, however, is invasive and lengthy, and there are logistical hurdles to overcome such as transporting the apheresis product to manufacturing locations.
Kyverna Therapeutics, a clinical-stage biotech, has developed a novel approach generating anti-CD19 CAR T cells for autoimmune indications from fresh whole blood rather than requiring apheresis, resulting in a shortened manufacturing cycle. A sample of the patient's blood is taken and processed using a special magnetic system. This system uses tiny magnetic beads to specifically pick out and activate the T cell. These cells are then modified using a lentiviral vector to introduce the CAR construct, grown in culture, and finally re-introduced into the patient. This method resulted in greater CAR-T potency at killing B cells, as well as reduced manufacturing time as compared to conventional manufacturing methods.
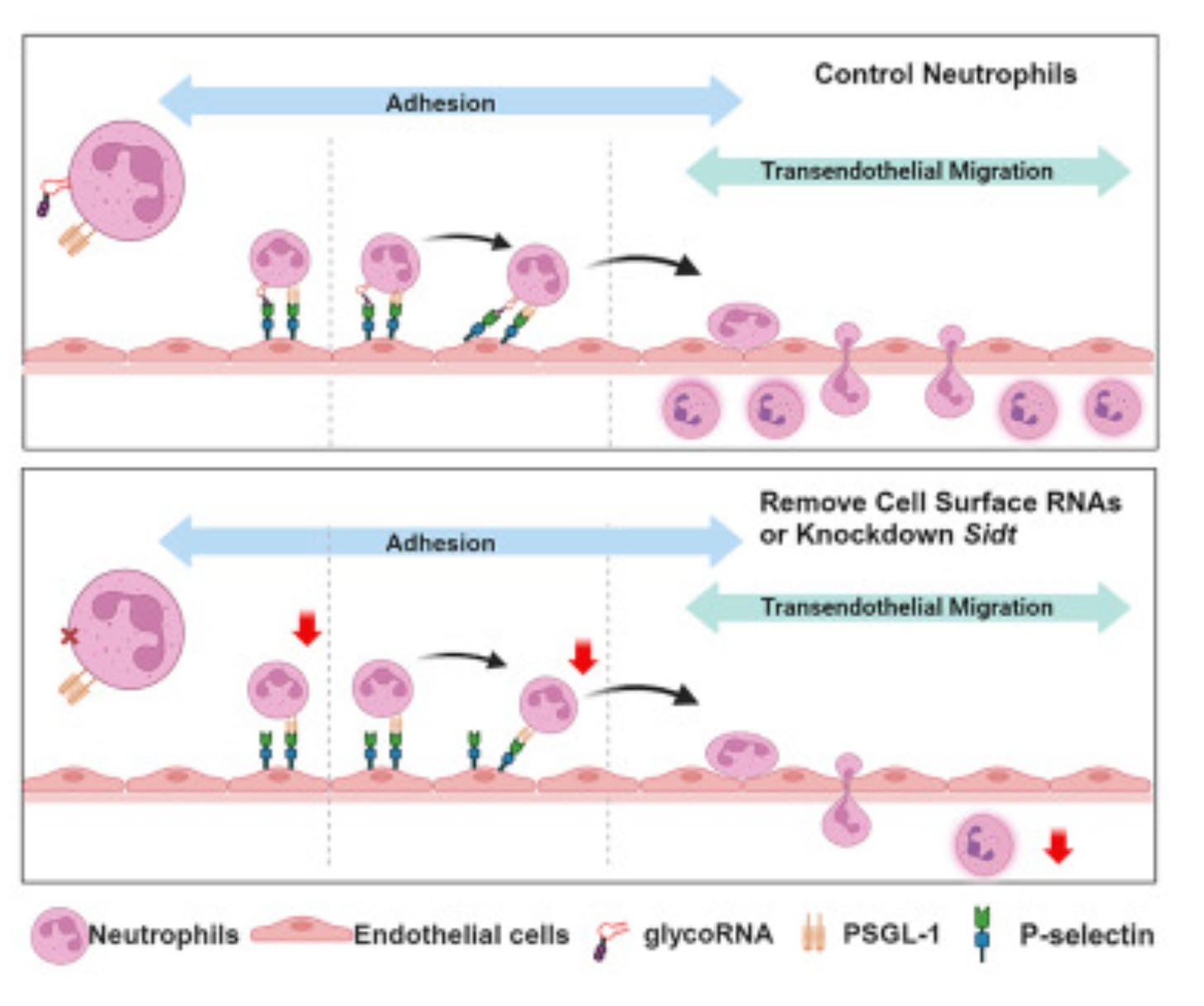
Cell surface RNAs control neutrophil recruitment [Zhang and Tang et al., Cell, 2024]
Why it matters: In recent years, work has shed light on the vast diversity of roles that RNA may have. In particular, a few papers have demonstrated the presence of extracellular RNAs, although their role was not well understood. Here, Zhang and Tang identify “glycoRNAs” on the cell surface of neutrophils, and that they are crucial for transendothelial migration, which is a critical step of the neutrophil response to inflammation. This work opens up a new avenue of biology, and brings to light an unorthodox aspect of RNA-enabled cell biology.Neutrophils must undergo a rapid series of complex interactions with vasculature and endothelium that enable their quick trafficking to sites of inflammation and infection. The authors found that certain cell surface RNAs (glycoRNAs, small RNAs that were mappable to noncoding transcripts and were subsequently glycosylated) were critical for neutrophil recruitment, and that an RNA transporter of the Sidt family was required for glycoRNA production and neutrophil-endothelial interaction. These RNAs were found on the extracellular neutrophil surface, and were recognized by the lectin (a carbohydrate binding protein) P-selectin. Eliminating said RNAs resulted in drastically reduced neutrophil margination, adhesion, and migration to sites of inflammation.
Of note, said GlycoRNAs were exceptionally stable, even though they were presented on the extracellular neutrophil surface. With a half-life of >24 hours, it appears that the glycoRNAs were protected from soluble RNAse – likely as a function of shielding proteins that haven’t been identified yet – while still able to maintain P-selectin binding. This data reveals a previously unknown dimension of immunobiology and represents a potential mechanism to modulate immune cell recruitment.
Notable Deals
POs / Reverse Mergers
CG Oncology prices upsized $380M IPO in hopeful sign for the IPO market - the biotech has a bladder cancer immunotherapy in phase III trials.
Tectonic Therapeutics reverse merges into gene-therapy-maker AVROBIO - this mechanism enables a private company (Tectonic) to go public by merging with an already public company (AVROBIO) which ceased development operations over the summer. This route can typically be shorter and more simple than the traditional IPO process.
Financings:
Motif Neurotech lands $18.7M Series A to fund depression-treating devices - the proceeds will be used to advance the company’s miniature brain pacemaker designed to precisely stimulate healthy circuit activity in the brain.
Cour Pharmaceuticals raises $105M to tackle autoimmune diseases - the biotech has developed polymer nanoparticles containing disease-specific antigens that serve to sensitive the immune response and contain overreaction to harmless antigens.
Cognito raises $35M for an Alzheimer’s treatment device - a headset that uses gamma frequency light and sound stimulation to combat the cognitive decline that’s the hallmark of Alzheimer’s.
Acquisitions:
Regeneron Pharmaceuticals to acquire 2Seventy Bio’s pipeline of cell therapies for $5M - the extreme restructuring has led 2Seventy retain a small commercial entity based on commercialisation of its CAR-T therapy Abecma. The rest of the pipeline will be acquired by Regeneron and a substantial portion of the staff will be let go. Increasing competition and dwindling commercial prospects for Abecma contributed to these changes.
Partnerships:
Elegen and GSK form a multi-year partnership around cell-free DNA production technology to enhance vaccine and therapeutic manufacturing - Elegen’s aim is to reliably generate long DNA sequences on demand. The collaboration includes upfront fees and purchase commitments to help progress Elegen’s technology.
Sanofi partners with Synthekine for $40M upfront to develop IL-10 receptor agonists for autoimmune and inflammatory diseases - Synthekine’s mission is to finally harness the true potential of cytokines in medicine by using novel biological approaches to overcome development and toxicity challenges.
Boehringer Ingelheim licenses multiple dark antigens from Enara Bio aiming to develop off-the-shelf immunotherapies for NSCLC. The licensing represents a major milestone in the strategic collaboration between the two companies announced in January 2021. Enara Bio used its EDAPT platform - which combines de novo bioinformatics databases with immunopeptidomics of tumor and healthy tissues and RNA in situ hybridization - to discover novel cancer-specific antigens from previously uncharted genomic ‘dark matter’.
What we listened to
In case you missed it
Zahra and Pablo recently interviewed Gevorg Grigoryan, CTO Generate:Biomedicines. The company has raised $700M to build an industrial generative biology platform. The team are looking for rising ML engineers interested in a 3 month internship. See the below:
Generate:Biomedicines | Open Positions
What we liked on socials channels
Events
Field Trip
Did we miss anything? Would you like to contribute to Decoding Bio by writing a guest post? Drop us a note here or chat with us on Twitter: @ameekapadia @ketanyerneni @morgancheatham @pablolubroth @patricksmalone