BioByte 065: RNA-editing therapies pick up steam, experts speak on protein design, decoding the autoantibody reactome, links between menopause and healthy aging, and more
Welcome to Decoding Bio, a writing collective focused on the latest scientific advancements, news, and people building at the intersection of tech x bio. Happy decoding!
This week is a busy one! From RNA editing to protein design to oocyte-replacement therapies, let’s get into it.
What we read
Blogs
Move over, CRISPR: RNA-editing therapies pick up steam [Nature, Feb 2024]
After decades of basic research into how to manipulate RNA, three therapies based on RNA editing are in clinical trials. RNA-editing techniques aim to change the sequence of RNA transcripts before they are translated into proteins. This is touted by some to be safer than DNA-editing, which causes permanent changes in the genome. Due to this transience, off-target effects from RNA-editing therapies may disappear quickly.
Protein design: the experts speak [Nature Biotech, Feb 2024]
Nature Biotech published a fascinating Q&A on the state of protein design with David Baker, Bonnie Berger, and Mohammed AlQuraishi. One of the biggest takeaways was that both DB and MA believe that despite advances in protein folding models like AlphaFold, the protein design field hasn’t actually discovered that much new about the underlying principles of protein folding. These models accurately predict the structure a given protein sequence will fold into, without knowing that much about the underlying physics/thermodynamics that governs the folding process.
One key trend in protein design is the need to integrate physics-based methods and knowledge into ML models. This is especially critical to solve problems in low-data regimes where data-hungry ML models will struggle, such as tasks involving non-canonical amino acids. The challenge is figuring out exactly how to integrate ML + physics-based models. For example, simply incorporating the Rosetta energy function (a measure of the free energy of a protein conformation) into protein structure prediction models like RoseTTAFold hasn't improved the accuracy of the model, so more sophisticated methods will be required.
Decoding the autoantibody reactome [Jaycox, Dai, and Ring, Science, 2024]
Autoimmunity has re-entered the spotlight in recent years as groups have continued to decipher key mechanisms driving immune responses and their role in disease. In particular, autoantibodies – antibodies that bind to native “self” antigens – have garnered focus as a major driver across a number of autoimmune diseases, such as Grave’s, rheumatoid arthritis, multiple sclerosis, and more. In this perspective, the authors shed light on more nuanced dynamics surrounding autoantibodies, and emerging technologies used to probe said molecules.
In particular, the authors posit that autoantibodies can be 1) clinically silent, or even 2) protective, depending on certain inciting factors or underlying disease dynamics. For example, during the COVID-19 pandemic, interferon-I (IFN-I) neutralizing antibodies were associated with an up to 200-fold increased risk of death from the virus. Interestingly enough, these autoantibodies appear to be clinically silent in most circumstances; however, it’s believed that 20% of all COVID-19 deaths were associated with IFN-I autoantibodies. On the flip side, autoantibodies may be protective - for example, in lupus, >50% patients have increased IFN-I signaling. The small subset of patients with IFN-I autoantibodies seem to have lower disease activity, believed to be secondary to reduction in pathologic IFN-I signaling. In a similar vein, autoantibodies against tumor-associated antigens (TAAs) have been associated with improved outcomes in cancer patients. These autoantibodies – and their targets – can provide critical information about targets and inform therapeutically relevant discovery campaigns.
Historically, one of the difficulties in pan-profiling of autoantibodies is that experimental methods for high throughput antibody detection are lacking; to date, autoantibodies have mainly been discovered via a biology-first approach, or via complex laboratory methods that lack throughput and scale. However, this is rapidly changing, as groups have focused on the development of next-gen antibody detection techniques. These include: phage immunoprecipitation sequencing (PhIP-seq) - a technique that encodes proteomic-scale peptide libraries for display on bacteriophage, which allows for immunoprecipitation for subsequent analysis via high-throughput DNA sequencing - and rapid extracellular profiling (REAP), which is is similar to PhIP-seq, but uses yeast to properly display full-length ectodomains of extracellular/secreted proteins.
The advent of these technologies opens possibilities to what the authors call “autoantibody-wide association studies” - akin to how DNA-sequencing enabled genome-wide association studies (GWAS, and has the potential to be another “layer” of molecular information that is clinically relevant. However, several challenges remain, including how 1) autoantibodies are quite rare, 2) they are not static across an individual’s lifetime or even disease progression, 3) they are often polyclonal mixtures of antibodies, and 4) antibodies can undergo their own alterations, such as isotype switching, posttranslational Fc modifications, and more. Despite the above, the evolution of our understanding of autoantibodies holds tremendous potential for biomedicine.
A guide to extend longevity by delaying menopause [Age1 and Carol Magalhaes, Libraries for the Future, February 2024]
The human lifespan has expanded significantly over the last few years but the lifespan of the ovaries has not matched pace. To contextualize with numbers, the life expectancy for women (in the US) is 80 years while the average age of menopause onset is 51. There has been a lot of discussion recently about studying menopause to study aging, specifically whether delaying menopause, which in turn would delay side-effects like migraines, brain fog, osteoporosis and more, could extend longevity. This post dives into the links between studying menopause and studying longevity, proposes theories on menopause’s impact to health, and suggests possible approaches for delaying menopause. It centers around two approaches—delaying menopause by targeting oocytes and treating menopause by targeting hormones.
A primer:
Women are born with all the oocytes they will ever have but they don’t mature until puberty
1000 oocytes are matured each month, only one of which is ovulated
Cells surrounding oocytes form follicles which produce progesterone, estrogen, and other necessary hormones
Quality of life diminishes after menopause onset suggesting a link between hormone production and other biological function
Studies have found women who live to 100 are 4x more likely to have had children in their 40s which proposes two hypotheses:
1) Healthy aging leads to delayed menopause
2) Delaying menopause leads to healthy aging
New DTC Marketing: Bringing our true customers along on our quest [Renny Gleeson & Chris Morrison, Rapport Bio, Feb 2024]
In this overview, the team at RA Capital describes how most pharma DTC marketing doesn’t bring pharma’s true customers alongside its mission. The authors state that given true customers are those that bring revenue dollars, then healthy people are the industry’s true customers. DTC ads typically pitch new therapies to patients, but don’t offer anything for healthy people.
Our industry’s lack of presence has led our customers to hear about us from others, leading to problematic regulation such as the IRA’s price controls on small molecules nine years after launch.
In February, Eli Lilly and Pfizer both released two ads that bring everyone along to their narrative around people “getting better” or “outdoing cancer”. I’d recommend watching both videos (linked in the blog post) which mark a new way of DTC marketing by bringing our true customers along the ride.
The World’s First Ethanol to Sustainable Aviation Fuel Production Facility [LanzaJet, Jan 2024]
US company LanzaJet is celebrating the grand opening of Freedom Pines Fuels Plant, the world’s first ethanol-based sustainable aviation fuel plant. The plant is commercial-scale produces jet fuel from ethanol, using microbes to perform much of the catalytic work. The microbe in question, Clostridium autoethanogenum is fed waste carbon monoxide and metabolises it into ethanol.
Academic papers
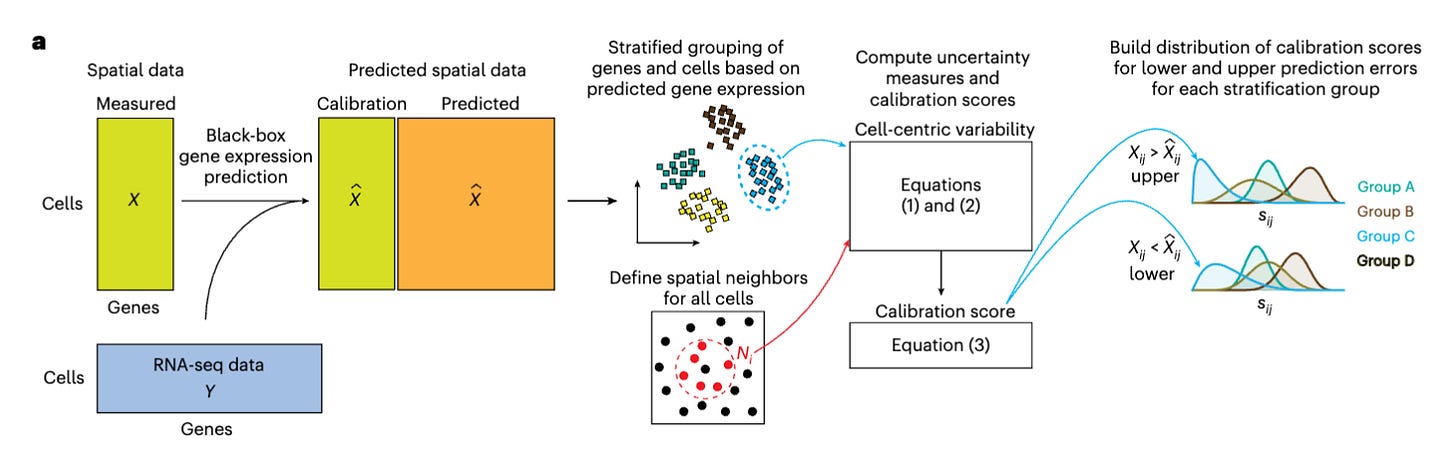
TISSUE: uncertainty-calibrated prediction of single-cell spatial transcriptomics improves downstream analyses [Sun et al., Nature Methods, February 2024]
Why It Matters: TISSUE addresses critical challenges in spatial transcriptomics by providing a robust method for quantifying prediction uncertainty, which is vital for the reliable interpretation of gene expression data. This advancement is expected to enhance the accuracy of biological insights gained from spatial transcriptomics studies, facilitating the identification of novel cell types and the understanding of complex tissue architectures. The ability to accurately predict gene expression with quantified uncertainty marks a significant step forward in the field, potentially accelerating discoveries in developmental biology, neuroscience, and disease pathology.The paper introduces TISSUE (Transcript Imputation with Spatial Single-cell Uncertainty Estimation), a framework designed to estimate uncertainty in spatial gene expression predictions and enhance downstream analyses in spatial transcriptomics. TISSUE employs conformal inference to generate well-calibrated prediction intervals for gene expression values across various datasets, significantly reducing false discovery rates in differential gene expression analysis and improving the performance of clustering and visualization techniques. By applying TISSUE to MERFISH spatial transcriptomics data of the adult mouse subventricular zone, the study identified neural stem cell lineage subtypes and developed regional classifiers specific to these subtypes.
Validation of biomarkers of aging [Moqri et al., Nature Medicine, February 2024]
Why It Matters: This research addresses a significant gap in aging research by providing a structured approach to validate biomarkers of aging, which are essential for advancing geroscience and developing interventions to extend healthspan. By establishing standardized validation processes and broadening the validation across diverse populations, the findings have the potential to catalyze the development of effective therapies for aging-related diseases, fundamentally shifting our approach to treating these conditions and enhancing the quality of life for the aging population.The paper provides a comprehensive review of the current efforts in the validation of biomarkers of aging, with a particular focus on 'omic'-based biomarkers. These biomarkers have the potential to predict aging-related outcomes and serve as surrogate endpoints in studies aimed at promoting healthy aging and longevity. However, there's a lack of consensus on validation methods, making their translation to clinical practice challenging. The authors discuss the predictive validity of these biomarkers in population studies, outline the challenges in ensuring their comparability and generalizability, and offer recommendations to facilitate their future validation. They emphasize the importance of systematic validation to accelerate the clinical translation of these biomarkers and their incorporation into gerotherapeutic trials.
Aging is identified as a primary risk factor for many chronic diseases and impairments, highlighting the potential of targeting the aging process itself to prevent multiple diseases simultaneously. The paper underscores the shift from biomarkers predicting chronological age to those focusing on biological age and health outcomes, reflecting the complex nature of aging. Despite advances, the acceptance of aging biomarkers in biomedical research remains limited, partly due to the absence of standard guidelines for their development, measurement, and validation.
Notable Deals
Approvals
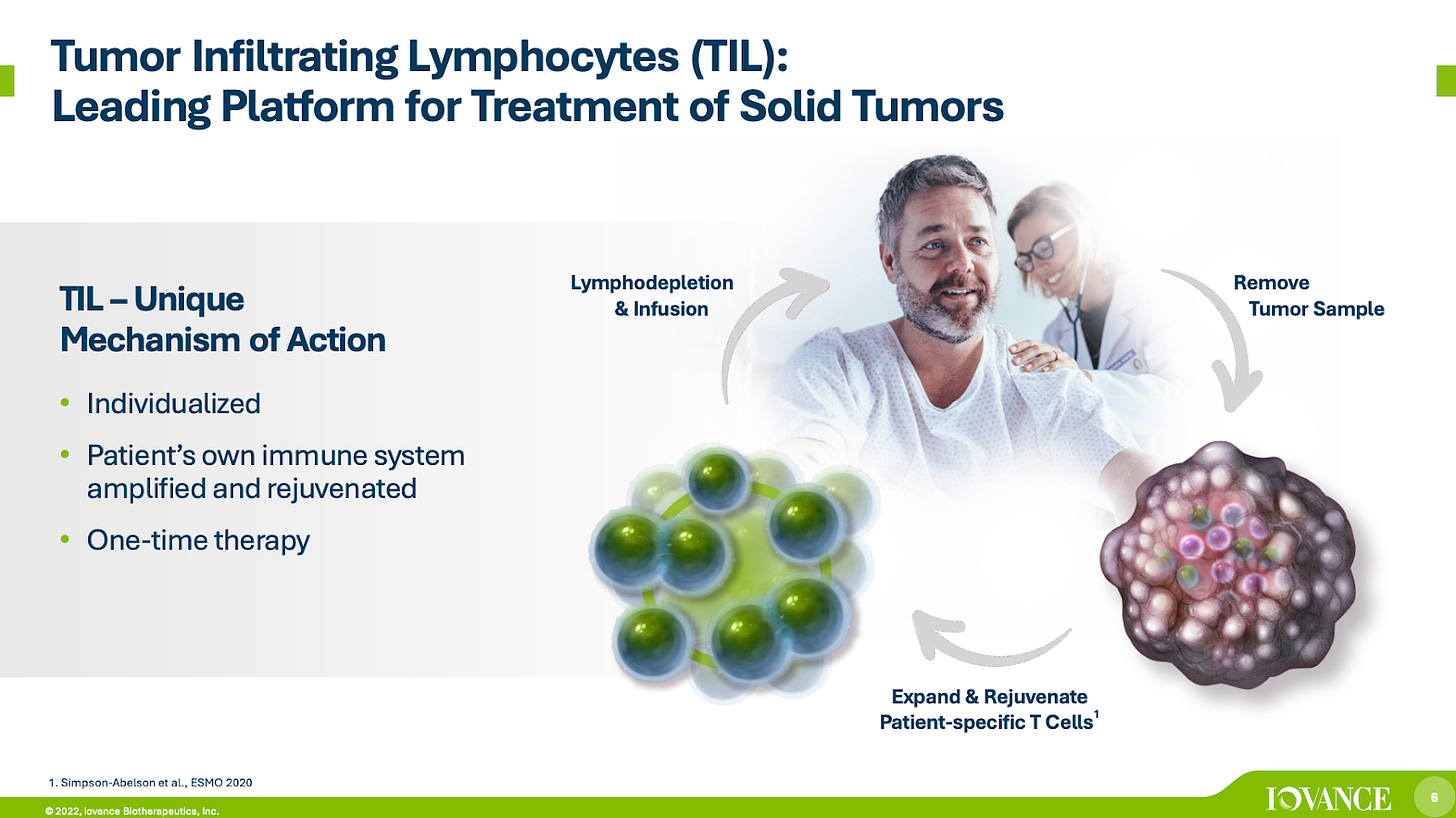
FDA approves the first cell therapy for solid tumors - Iovance’s TIL (Tumour Infiltrating Lymphocyte) therapy has received accelerated approval for advanced melanoma. Despite being discovered and proven to work in the 1980s by Steven Roseburg, cell therapies such as TILs have struggled in solid tumors. Iovance’s Amtagvi (Lifileucel) is a first-in-technology approval. However, despite their efficacy, TILs still evoke toxicity due to the IL-2 treatment and harsh chemotherapy required on administration.
Source: Iovance Biotherapeutics Company Presentation
Financings
Owkin and Ex-Google Deepmind scientists launch Bioptimus with $35M in seed funding - the French venture to build foundation models at various scales of biology. The models will be trained on massive amounts of biological and multimodal data across scales, aiming to make sense of biology’s complexity. Owkin will contribute its data generation capabilities with federated global access to multi-modal patient data sourced from leading academic hospitals worldwide.
Firefly Bio launches with $94M with a novel take on ADCs - Carolyn Bertozzi (Nobel Prize 2022) is a cofounder of the new biotech that is using sophisticated chemistry for innovative linker strategies to design and optimize all the different elements of the ADC. Firefly’s approach is to attach a degrader (instead of a cytotoxic payload) to the antibody. The idea is that degraders act intracellularly and so may spare more healthy tissue and have an improved therapeutic index as compared to traditional ADCs. The company is backed by Versant Ventures and MPM Capital.
Freenome raises $254M to advance its early cancer detection tests - the funding will enable the company to advance its pipeline of single-cancer and tailored multi-cancer early detection tests built on its multi-omics platform. Freenome is also evaluating its tests for Minimum Residual Disease with its biopharma partners. The round was led by Roche.
Profound Bio raises $112M Series B to progress its pipeline of ADCs
Partnerships
Intellia Therapeutics and ReCode Therapeutics partner to develop genetic medicines for cystic fibrosis - the companies hope that Intellia’s “DNA writing” technology and ReCode’s methods for delivering genetic medicines to specific organs will together help develop a novel in vivo gene therapies for lung diseases. Intellia will be responsible for the design of the editing strategy and research-grade components for the investigational therapies. ReCode will lead the subsequent preclinical and clinical development.
Astellas and Kelonia partner to develop “in vivo” cell therapies - the two companies are combining their technologies to develop medicines that introduce a gene to immune cells, modifying them inside the body rather than the lab. Astellas will pay $40M upfront to develop one initial program and could add $35M more if it exercises an option on another.
What we listened to
What we liked on socials channels
Events
San Francisco founders! We’re co-hosting a small dinner for those building companies in the AI x Bio ecosystem in March. Have a few spots left, shoot us an email at decodingbio@gmail.com if interested.
Field Trip
Did we miss anything? Would you like to contribute to Decoding Bio by writing a guest post? Drop us a note here or chat with us on Twitter: @ameekapadia @ketanyerneni @morgancheatham @pablolubroth @patricksmalone