BioByte 067: DNA foundation models, neural multi-task learning for drug design, non-invasive methods for brain amyloid clearance, clinical trial architectures for precision medicine
Welcome to Decoding Bio, a writing collective focused on the latest scientific advancements, news, and people building at the intersection of tech x bio. Happy decoding!
It’s March? That was fast. We’re keeping it short and sweet this week, but one for the neuro nerds… Read on!
What we read
Blogs
Evo: DNA foundation modeling from molecular to genome scale [Arc Institute, February 2024]
Evo is a groundbreaking biological foundation model designed for understanding and generating DNA, RNA, and protein sequences, offering prediction and generative design capabilities across molecular to genome scales. Developed by researchers from Arc Institute, Stanford, and TogetherAI, Evo utilizes a StripedHyena architecture for enhanced processing of long genomic sequences. This work shines in zero-shot gene essentiality testing and cross-modal biological predictions, setting a new benchmark for AI's role in biological sciences.
One of the most striking capabilities of Evo is its ability to perform multimodal design tasks to generate novel CRISPR Systems, “a task that requires creating large functional complexes of proteins and ncRNA (non-coding RNA), and is out of reach for existing generative models. Typically, discovering new CRISPR systems requires searching through natural genomes for similar sequences that were literally taken from an organism. Instead, Evo enables a new approach to generating biological diversity by sampling sequences directly from a generative model, an exciting frontier for creating new forms of genome editing tools.”
Access Evo via this GitHub repo.
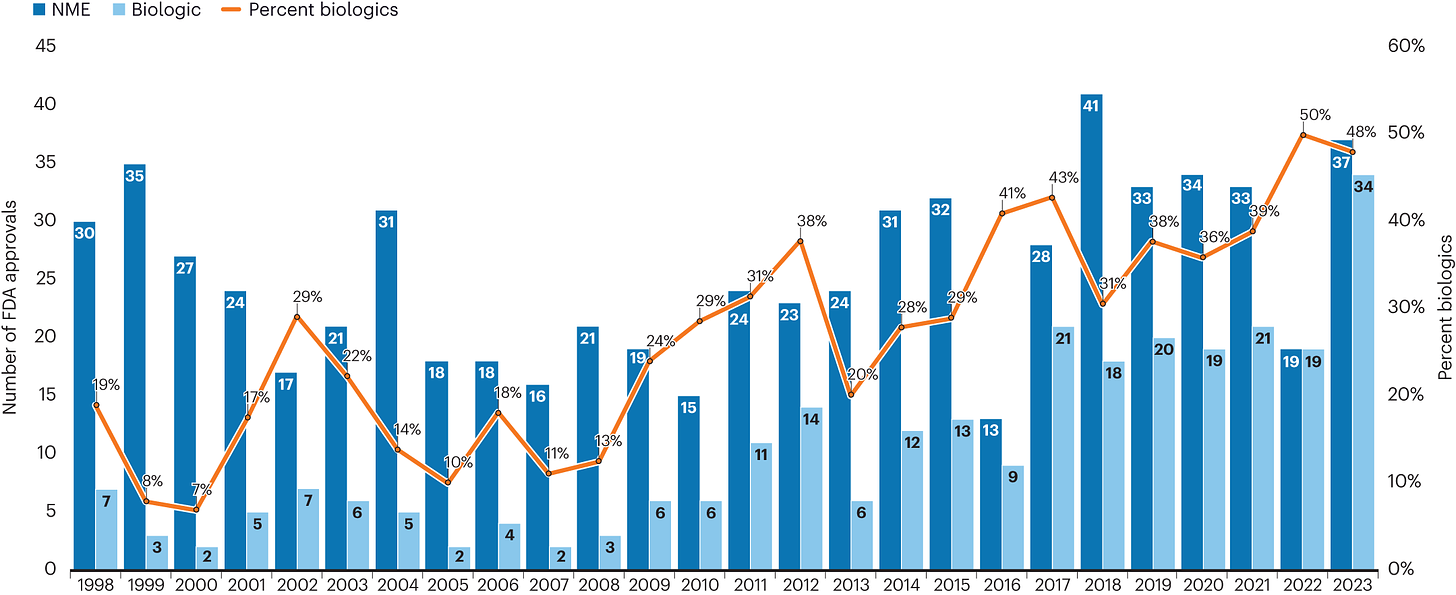
Fresh from the biotech pipeline: record-breaking FDA approvals [Senior, Nature Biotechnology News, February 2024]
The FDA marked a significant milestone in 2023 with a record 71 new drug approvals, including groundbreaking gene therapies and the first CRISPR–Cas9-edited therapy. This achievement, reflecting both innovation in biotechnology and a proactive regulatory stance, signals an encouraging landscape for future drug development. Among the highlights were treatments addressing a wide range of conditions from rare diseases to prevalent health concerns like Alzheimer’s and RSV, underscoring the FDA's commitment to advancing healthcare solutions across the spectrum of patient needs.
Finding the rare disease needle in the healthcare data haystack [Vera Mucaj, Chief Scientific Officer at Datavant, Medium]
In the labyrinth of healthcare data, rare diseases present a unique challenge with fragmented information that hampers timely diagnosis and treatment. The article outlines a visionary approach where real-world data and advanced analytics emerge as beacons of hope. By knitting together vast, disparate data sources into a cohesive fabric, the potential to uncover hidden patterns and accelerate the diagnostic process is unprecedented. Emphasizing patient privacy, the article advocates for a dual strategy: enhancing data linkage while ensuring robust privacy safeguards. The ultimate goal is a dynamic, evolving dataset that not only aids in the discovery of rare diseases but also propels the development of groundbreaking treatments, promising a future where the journey from symptoms to solutions is dramatically shortened.
All models are wrong and yours are useless: making clinical prediction models impactful for patients [Florian Markowetz, Nature Precision Oncology Comment, Feb 2024]
I think your model is useless. How would I know? I don’t even know who you are. Well, it is a bet. A bet I am willing to take because the odds are ridiculously in my favour.
Florian notes his frustration with the lack of translatability of academic success of clinically relevant models to clinical impact, even in his own published work. He makes several observations from his last two decades in the field:
Success in academia is not the same as success in the clinic. Implementation, which is what would contribute to clinical impact, is not rewarded by academia. But publishing new models, always is. Incentives are misaligned.
Successful models use data that are available in routine practice. The academic view of what is important (multiomics, spatial) is at odds with clinical reality where clinicians might only have access to an H&E slide and DNA sequences.
Successful models are linked to actions. Models are useless without a prognostic score or intervention recommendation.
Successful models are implemented outside of centres of excellence. Models that are only in Cambridge, Stanford or Zurich are going to have narrow utility.
Success in the clinic is hard earned. Hospitals are conservative, doctors overworked, health systems underfunded. You will need substantial evidence of the usefulness of the model before hospitals take into considerations your academic insight on board.
Academic papers
Neural multi-task learning in drug design [Allenspach et al., Nature Machine Intelligence, February 2024]
Why it Matters: The significance of multi-task learning in drug design stems from its potential to enhance early-stage drug discovery by efficiently assessing compounds for specific properties. The vast chemical space of drug-like molecules poses a challenge in identifying effective treatments, and MTL offers a promising approach to navigate this complexity. By enabling the prediction of binding affinities for multiple proteins simultaneously, MTL models can leverage similarities across datasets to improve overall predictive capabilities.
The article discusses neural multi-task learning (MTL) in drug design, focusing on its principles, applications, and challenges. The discussion highlights MTL's potential to improve predictive models by leveraging shared information across multiple tasks, specifically in predicting protein–ligand binding affinities. The authors introduce a framework for categorizing MTL models based on architecture and discuss the challenges in defining suitable molecular representations and learning tasks.
The article underscores the importance of MTL in drug design by articulating the potential to address chemical space using MTL approaches, and outlining the need for efficient drug discovery processes. MTL's ability to harness shared knowledge across tasks and its application in protein–ligand binding affinity prediction highlight its potential to significantly advance drug design. This approach not only enhances predictive accuracy but also contributes to the broader goal of discovering effective treatments more efficiently.
Multisensory gamma stimulation promotes glymphatic clearance of amyloid [Murdock et al, Nature, Feb 2024]
Why it matters: Li-Huei Tsai and Ed Boyden’s lab demonstrate that multisensory audio-visual sensory stimulation in the gamma frequencies can promote glymphatic-mediated clearance of amyloid plaques in the brain of a mouse model of Alzheimer’s disease. These findings suggest a potential non-invasive therapeutic strategy for leveraging the brain’s waste clearance system to treat neurodegenerative disorders.
The glymphatic system plays a critical role in clearing metabolic waste products like amyloid-β from the brain. Previous studies found that inducing gamma frequency (40 Hz) neural oscillations via optogenetic or sensory stimulation reduced amyloid pathology in mouse models of Alzheimer's disease (Cognito Therapeutics was founded based on these findings, and is currently in clinical trials), but the mechanisms were unknown. The authors hypothesized that multisensory audio-visual gamma stimulation may enhance glymphatic clearance of amyloid by increasing the influx of cerebrospinal fluid (CSF) into the brain and the efflux of interstitial fluid (ISF) containing waste products.
Using fluorescent tracer imaging, two-photon microscopy, chemogenetics, and single-nucleus RNA sequencing, the authors found that multisensory 40 Hz gamma stimulation promoted the influx of CSF into the cortex and efflux of ISF in a mouse model of Alzheimer's disease, resulting in the clearing of amyloid deposits from the brain (image below shows amyloid deposition as a function of frequency of sensory stimulation, with the greatest clearance occurring at 40 Hz). The increased CSF influx was associated with greater polarization of the water channel aquaporin-4 on astrocytic endfeet, dilation of meningeal lymphatic vessels, and increased arterial pulsatility - factors that regulate glymphatic flow. This work suggests potential therapeutic strategies for Alzheimer's disease and other protein misfolding disorders involving recruitment of the glymphatic clearance system through neuromodulation of specific brain rhythms.
Crym-positive striatal astrocytes gate perserverative behavior [Ollivier et al., Nature, 2024]
Why it matters: Historically, neurons were believed to be the cells in the brain solely responsible for driving behavior. In this paper, the authors demonstrate that a subpopulation of astrocytes – a type of glial, or supportive cell, in the brain – contribute to repetitive behaviors (perserveration). Additionally, they identify a gene, Crym, that encodes for the µ-crystallin protein and is intimately linked to such repetitive behavioral patterns. This work adds to the emerging body of literature implicating glial cells in homeostasis and neuropsychiatric disorders.
Astrocytes have been well established to provide supportive functions in the brain milieu - regulating blood flow, maintaining extracellular fluid ion concentrations, synaptic function, and more. Recent work in this area has demonstrated that specialized astrocytes exist in unique brain regions, and establish associated locoregional neural circuits. However, their exact role in the progression of various diseases has remained unclear. In this paper, Ollivier et al., identify a unique population of astrocytes in the striatum – a region of the brain involved in motor control, emotion, etc. – that are marked by expression of the gene Crym, and regulate motor movements.
Crym encodes for the protein µ-crystallin, which has been previously associated with various neurocognitive disorders. The authors probed the expression of µ-crystallin and found that it was densely expressed in the astrocytes of the striatum, and not neurons. After knocking down Crym in mice, the authors found that doing so had no effect on motor control or anxiety, but induced a profound increase in perseveration - repetitive behaviors that have no purpose. Perseveration is noted to be a marked component of a variety of neurocognitive disorders. Subsequently, the authors explored the underlying mechanism driving manifestations of Crym loss in the striatum, and found that this influenced the ratio of excitatory and inhibitory neuronal signals, leading to perseveration. This work elucidates a hitherto unknown mechanism of astrocytes mediating neurological function, and represents new potential means for therapeutic management.
New clinical trial design in precision medicine: discovery, development and direction [Nature Signal Transduction and Targeted Therapy, March 2024]
Why it matters: The traditional one-size-fits-all approach to clinical trials is not sufficient for the personalized approach surrounding precision medicine and genetic makeup of individuals. This paper discusses innovative clinical trial designs, such as basket, umbrella, and platform trials, which are essential for evaluating targeted therapies and addressing the diverse needs of patients with various genetic alterations.
This review paper explores innovative clinical trial designs tailored to precision medicine. It emphasizes the importance of biomarker-guided trials, such as basket, umbrella, and platform trials, in addressing patient heterogeneity and enhancing treatment personalization. These designs facilitate the efficient exploration of targeted therapies. The three main trial designs discussed are highlighted below, the article itself gets into the specific variations of each trial including what disease types they may be best suited to study.
Basket Trials: These trials investigate the efficacy of a single drug or intervention across multiple diseases or conditions that share a common biomarker. For example, a basket trial might test a targeted therapy for a specific genetic mutation across different types of cancer. This approach allows for the rapid assessment of a drug's potential across a variety of clinical contexts.
Umbrella Trials: In contrast to basket trials, umbrella trials focus on a single disease but test multiple targeted therapies simultaneously. Each therapy targets a different biomarker or molecular pathway within the disease. For example, an umbrella trial in lung cancer might evaluate several different treatments, each targeting a specific genetic alteration found in subgroups of lung cancer patients. This design enables the identification of the most effective therapy for each patient's unique disease profile.
Platform Trials: These are dynamic trials that allow for the continuous addition of new therapies and the removal of ineffective ones over time. Platform trials are designed to be adaptable and efficient, with a common infrastructure that can support multiple simultaneous investigations. This approach is particularly useful in rapidly evolving fields like oncology, where new treatments are constantly being developed.
Notable Deals
Backed by MIT’s Andrew Lo, Quantile Health raises $6M to bolster gene therapy access.
Sionna raises another $182M to challenge Vertex in cystic fibrosis.
Startup Alumis raises $259 million as it prepares for psoriasis trial.
In case you missed it
Last summer we hosted our inaugural AI x Bio Summit at the NYSE. We’re planning the 2024 edition which will likely be in the Bay Area. If you have any ideas for the program, would like to speak at the event or attend it do message us (twitter/linkedin/email open).
Basecamp’s Medium Post: “Why we’re so excited about the Arc Institute’s new Evo model.”
Field Trip
Did we miss anything? Would you like to contribute to Decoding Bio by writing a guest post? Drop us a note here or chat with us on Twitter: @ameekapadia @ketanyerneni @morgancheatham @pablolubroth @patricksmalone