BioByte 069: AI learns the meaning of life, the dysfunctome, a $400M launch for inflammatory drugs, Nvidia's 'woodstock' and engineering crops to save the planet
Welcome to Decoding Bio, a writing collective focused on the latest scientific advancements, news, and people building at the intersection of tech x bio. Happy decoding!
Its Q1, you’re busy, we’re busy… but he’s your weekly BioByte.
What we read
Blogs
Seeds of a wild idea [Direct Driller Magazine, March 2024]
“Wild plants could hold the genetic key to a step change in productivity across major agricultural crops. Tech Farmer visits the labs of a new start-up that’s sifting through the genomes.”
AI is learning what it’s meant to be alive [Carl Zimmer, New York Times, March 2024]
Zimmer profiles the potential impact of AI on biology in this captivating piece, starting with a story of how it took AI six weeks to naturally discover Norn cells, a kidney cell responsible for producing erythropoietin when oxygen levels are low, when it took humans 134 years. Notably, while the AI was trained on tons of cellular data, it did not have predefined knowledge on cell type and function yet managed to classify 1000+ cell types. Similar foundation models can be used to understand gene behavior in different cell types and, taken one step further, can help predict interventions for specific diseases. The article discusses several applications of foundation models to biology including the Universal Cell Embedding model which can classify cells based on their genetic activity—important for developmental biology and relating such knowledge to other species. Not all rosy, Zimmer touches on the challenges involved including need for data, ethics, and the limitations of such models.
The Road to Biology 2.0 Will Pass Through Black-Box Data [Michael Bronstein, Towards Data Science, 2024]
Michael Bronstein and Luca Naef have published a short piece exploring the potential of AI in biology, the characteristics of biological problems that are most conducive to being solved by AI techniques and contend that the key to success of ML approaches in biology will be the generation and use of ‘machine-friendly’ not ‘human-friendly’ data. That is, high-throughput data that is not necessarily ‘valuable.’ The article sets the scene by introducing the complexities of ML in biology: biological systems operate across temporal and spatial scales, biological datasets are often subject to batch effects or artefacts.
Covid, 4 years on [Eric Topol, Ground Truths, 2024]
March 11th 2024 marked the 4-year anniversary of when the WHO declared Covid a global pandemic. The global excess mortality has reached about 30 million deaths attributable to Covid.
In the US, updated data on partisan gap death rates show continued divergence. Since the vaccines were first administered, the death rates continue to worsen in counties with >70% republican voters compared with <30%.
A few other interesting tidbits:
BA.2.87.1 seems like it is the major “Omicron-like” event with no sign it is more immunoevasive or transmissible.
Boosters halve the death rate in people 65 and older
Prior covid vaccination protects from cardiovascular issues related to covid infection (deep vein thrombosis and pulmonary embolism, heart attacks, strokes, and heart failure)
Academic papers
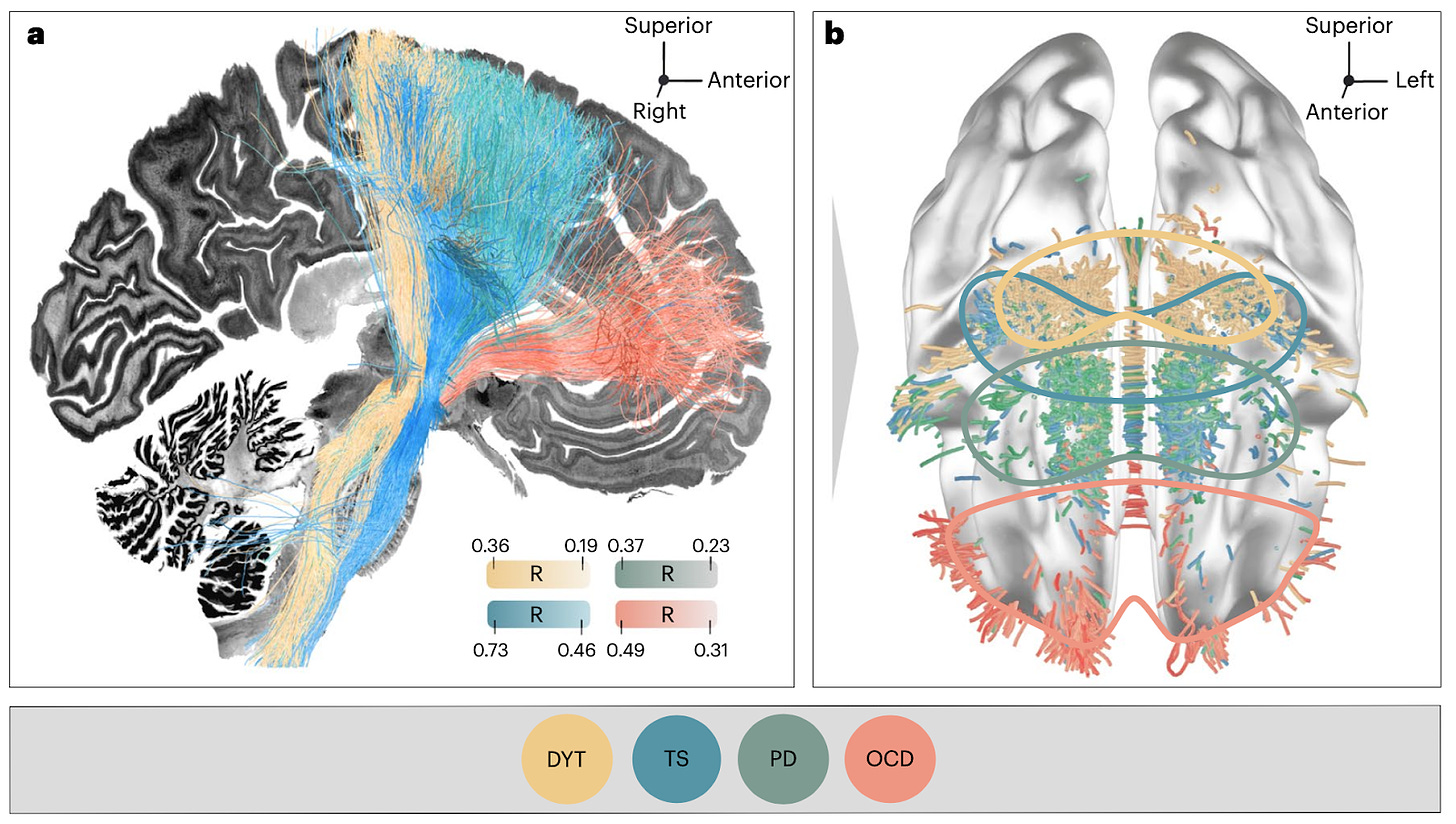
Mapping dysfunctional circuits in the frontal cortex using deep brain stimulation [Hollunder et al., Nature Neuro, Feb 2024]
Why it matters: This study provides a novel framework for mapping dysfunctional brain circuits across neuropsychiatric disorders by integrating deep brain stimulation outcomes with brain connectivity data. Andreas Horn’s lab identified a topographic organization of dysfunctional circuits in the frontal cortex and validated these circuits as therapeutic targets. By illuminating the "dysfunctome" and establishing the integration of neuromodulation with connectomics, this work represents a significant advance towards precision psychiatry.
Neural circuits in the frontal cortex - the area of the brain right behind the forehead - play a critical role in many neuropsychiatric diseases. The spatial distribution of different neural circuits within the frontal lobes across diseases and patients, and how these spatially separate circuits can be selectively targeted with brain stimulation to treat disease, is an area of active focus.
Andreas Horn’s lab studied 534 deep brain stimulation (DBS) electrodes implanted to treat four different brain disorders - dystonia, Parkinson's disease, Tourette's syndrome, and obsessive-compulsive disorder (OCD). By analyzing which brain connections were modulated for optimal therapeutic response across these disorders, they found the frontal cortex segregated into distinct circuits that had become dysfunctional in each disorder (figure below). The dysfunctional circuits were topographically arranged from occipital to frontal, ranging from:
Interconnections with sensorimotor cortices in dystonia
Primary motor cortex in Tourette's
Supplementary motor area in Parkinson's
Ventromedial prefrontal and anterior cingulate cortices in OCD
The findings highlight the integration of DBS with brain connectomics as a powerful approach to explore links between brain structure and functional impairments in humans.
The models were validated by prospectively reprogramming brain stimulation parameters in 3 patients to specifically target the identified neural circuits, which lead to symptom improvements. This approach highlights the importance of integrating DBS with brain connectomics as a powerful precision medicine approach to improve the therapeutic efficacy of neuromodulation.
Intrathecal bivalent CAR T cells targeting EGFR and IL13Rɑ2 in recurrent glioblastoma: phase 1 trial interim results [Bagley et al., Nature Medicine, 2024]
Why it matters: Glioblastoma (GBM) remains the most common and most aggressive brain cancer, with an extremely poor prognosis (< 1 year). GBM is a highly recurrent disease; given the resilience of the disease, groups have tried to understand whether a living therapy, such as CAR-T, would improve outcomes in these patients. Here, Bagley and colleagues developed CAR-T cells that target both the epidermal growth factor receptor (EGFR) and the tumor associated antigen interleukin-13 receptor alpha 2 (IL13rɑ2) and treated six patients with these. All patients had short-term tumor regression, while 50% of patients did not have any subsequent progression (currently within six months of treatment).
Given the heterogeneity of glioblastoma, and the lack of efficacy in monovalent targeting, the authors decided to develop a bivalent CAR-T therapeutic against the aforementioned targets. T-cells were transduced with a bicistronic lentiviral vector containing a chimeric scFv targeting the EGFR epitope 806, and the tumor associated antigen IL13Rɑ2. Of note, the EGFR epitope is a conformational epitope that is largely only targetable in the context of EGFR overexpression and/or in the context of certain EGFR extracellular domain mutations – both of which are found to be a common occurrence in GBM.
After intrathecally dosing these CAR-Ts in patients, the authors reported that these cells reduced tumor size and enhancement (a proxy for vascularity and cellular replication) in all six patients. While none met criteria for an objective clinical response, tumor shrinkage and stable disease was achieved for some of these patients Additionally, although an early-onset acute neurotoxicity was observed in patients, there were no grade 4 or 5 toxicities observed. While long-term outcomes remain to be seen, this work is exciting as it helps set the foundation for optimizing CAR-Ts for glioblastoma, and provides hope that we may still have a chance at treating this disease.
Notable Deals
M&A:
AstraZeneca splashes $2B to join the radiopharmaceutical space - The British pharma has struck a deal with Fusion Pharmaceuticals which also includes up to $400 million in contingent value rights. The companies first started working together more than three years ago to develop combination therapies. Fusion’s lead candidate targets PSMA (enriched in mCRPC) and is currently in Phase II. The company also has a suite of actinium-based radio-conjugates for solid tumors such as a Phase I EGFR program. The deal not only brings a rich pipeline, but importantly, key manufacturing and supply chain capabilities - crucial factors to the success of any radiopharmaceutical drug development campaign.
Financings:
Mirador Therapeutics raises $400M to develop new medicines for inflammatory diseases - The biotech is lead by the same team that brought Prometheus Therapeutics to its $10.8B buyout by Merck last year. This time, the team are working again on inflammatory diseases, using their Mirador360 platform built to harmonize millions of patient molecular profiles to discover and validate genetic associations to immune-related disorders. The capital also enables the team to acquire or license further inflammatory assets of interest.
Clasp Therapeutics launches with $150M from Catalio, Third Rock, Novo to develop -cell engager bispecifics against common driver mutations of cancer that contribute to immunotherapy resistance. The start-up is born out of work from Bert Vogelstein and Drew Pardoll and the funding will enable the gathering of early proof-of-concept clinical data. Instead of targeting cell-surface proteins, Clasp’s engagers will target mutated proteins found inside cancer cells, this may enable the therapeutics to avoid harming healthy tissue.
Zephyr AI Raises $111 Million in Series A Financing - the company is developing fast and explainable artificial intelligence solutions to democratize precision medicine. Zephyr aims to do this by combining machine learning expertise with healthcare partnerships that enable access to real-world clinico-genomics datasets. The round saw participation from Eli Lilly.
Biswas Family Foundation, Milken Institute Announce $15 Million in Funding for Research at the Intersection of AI and Health.
Capstan raises $175M Series B for in vivo CAR-T - this technology promises to produce more affordable therapies that are less complex to manufacture and thus more widely accessible. The technology uses an mRNA payload to target CD8+ T cells and is based on work by CAR-T pioneers Carl June and Drew Weissman The company has backing from several large pharma companies, highlighting industry interest (Johnson & Johnson Innovation, Bristol Myers Squibb, Eli Lilly, Leaps by Bayer, Novartis Venture Fund and Pfizer Ventures).
Engrail raises a $157M series B to further its neuropsychiatric pipeline - following the recent neuropsych M&A trend of pharma interest, the biotech was able to accelerate its raise. The capital extends the company’s cash runway to mid 2026 with a focus on its generalized anxiety asset ENX-102 that is currently in Phase II testing.
Profluent Bio announces $35M in additional funding led by Spark Capital to use AI as an interpreter to decode the language of life.
What we listened to
In case you missed it
What we liked on socials channels
Field Trip
Did we miss anything? Would you like to contribute to Decoding Bio by writing a guest post? Drop us a note here or chat with us on Twitter: @ameekapadia @ketanyerneni @morgancheatham @pablolubroth @patricksmalone