BioByte 072: DNA printing, edible neuromodulatory electronics, AI agents empower biomedical discovery, scRNAseq supports successful drug targets, $100M seed round for gene editing delivery newco
Welcome to Decoding Bio, a writing collective focused on the latest scientific advancements, news, and people building at the intersection of tech x bio. Happy decoding!
What we read
Blogs
AminoX: Making Better Protein Drugs, Quicker and Cheaper [Wyss Institute, 2024]
Northpond Ventures has funded Wyss Institute’s AminoX project - the aim is to develop a platform for the rapid creation and integration of non-standard amino acids (nsAA) into protein drugs, endowing therapeutics with new functions more easily. Such amino acids have been incorporated in breakthrough drugs such as Ozempic.
The problem: although proteins offer an effective way to treat many diseases, in their natural form these proteins can cause toxicities or have other non-ideal properties for therapeutics such as short half-life.
The solution: AminoX platform uses a novel approach to incorporate nsAAs into proteins by generating functional intermediates that can effectively hand over their nsAAs directly to the ribosome in a scalable and specialized cell-free process. The platform also uses machine learning to identify new nsAA designs that may endow target proteins with new functions.
Research Leaders’ Playbook [Ben Reinhardt, Speculative Technologies, April 2024]
Ben has put together a comprehensive guide for driving innovation and excellence in research organizations. At its core, the playbook emphasizes the importance of building a thriving research culture—one that cultivates psychological safety, encourages interdisciplinary collaboration, and recognizes creative thinking. He outlines strategies on essential skills needed to be a great research leader covering topics including how to build a research culture, assembling a talented team, aligning research with organizational objectives, scaling capabilities and impact, communicating insights, and navigating ethical and regulatory considerations. It’s a great read for anyone working on coordinated research initiatives such as FROs, ARPA-H or DoD programs, or accelerators and foundations. We found this playbook particularly actionable and relevant to various stages of research organizations.
The Wizardry and Prophecy of DNA Printing [Kat Baney, Ideas Matter, April 2024]
The blog post delves into the dichotomy between two archetypes in the realm of technological advancement: the Wizard and the Prophet. The Wizard embodies a mindset of innovation and experimentation, believing that science and technology will solve humanity's problems, while the Prophet advocates caution and ethical considerations, warning against the potential misuse or unintended consequences of new technologies.
This dynamic is exemplified through the contrasting perspectives of Norman Borlaug, the agronomist behind the "Green Revolution," and William Vogt, an ecologist advocating for sustainability and conservation. The post explores how this dichotomy plays out in the field of synthetic DNA production, with individuals like Emily Leproust and Keoni Gandall embodying the Wizard archetype, pushing for widespread access and innovation in DNA printing, while others, like Kevin Esvelt, adopt a Paladin role, combining innovation with safeguards against potential risks.
The post highlights the importance of balancing innovation with responsible stewardship in the rapidly evolving landscape of biotechnology, emphasizing the need for collaboration between Wizards, Prophets, and Paladins to navigate the ethical, security, and regulatory challenges inherent in technological progress.
Academic papers
Single-cell RNA sequencing of human tissue supports successful drug targets [Dann et al., MedRxiv, April 2024]
Why it matters: A recent preprint from a collaboration across academia, biotech, and large pharma provides evidence that single-cell RNA sequencing data can be leveraged to identify drug targets that are significantly more likely to succeed in clinical trials. The approach is complementary to using human genetics data and can prioritize a larger set of clinically effective targets. This highlights the value of incorporating single-cell data into drug discovery pipelines to increase success rates.
A new preprint from a collaboration between the Teichmann Lab at the Sanger Institute, Ensocell Therapeutics, and Sanofi provides compelling evidence for how single-cell RNA sequencing of human tissue can successfully identify drug targets. The authors analyzed single-cell RNA sequencing data from 30 diseases and 13 tissues to identify genes with cell type specific expression in disease-relevant cell types or specific overexpression in diseased compared to healthy tissues. They found that these genes were significantly more likely to be targets of clinically successful drugs, with a 3-fold higher probability of passing safety and efficacy trials compared to genes without such evidence. By comparing to drug target associations from human genetics, they show that single-cell data can prioritize a distinct and larger set of therapeutic targets, demonstrating its complementary value in drug discovery pipelines to increase clinical success rates and accelerate the development of new treatments.
Going forward, developing more sophisticated analysis methods for scRNA-seq data, such as improved cell type annotation, detection of disease-associated cell states, cell-cell interactions, regulatory networks, etc all may further increase the probability of successful translation of therapeutic targets identified in scRNA-seq data.
Edible electronics to treat the brain [Khalil Ramadi, Science, April 2024]
When it comes to neuromodulation, there is a fundamental trade off between invasiveness and specificity. To address this issue, the author sought to use direct stimulation of neuronal endings in the gastrointestinal tract.
The logic of this is that the GI tract is i) home to the largest number of neurons in the body outside the brain (the enteric nervous system) and ii) has a lumen which is technically outside the body. This means that interfaces with enteric neurons without penetrating the epithelium are noninvasive.
Electrical neuromodulation is not novel. The invasive gastric electrical stimulation systems are FDA approved for gastroparesis. The 2023 paper proposed the concept of FLASH, an ingestible bioelectronic that can modulate hormone secretion. It showed modulation of ghrelin (a hunger promoting hormone) released by the stomach to increase hunger. Thus, clinical indications include eating disorders and chronic nausea. FLASH is currently being spun out for clinical use.
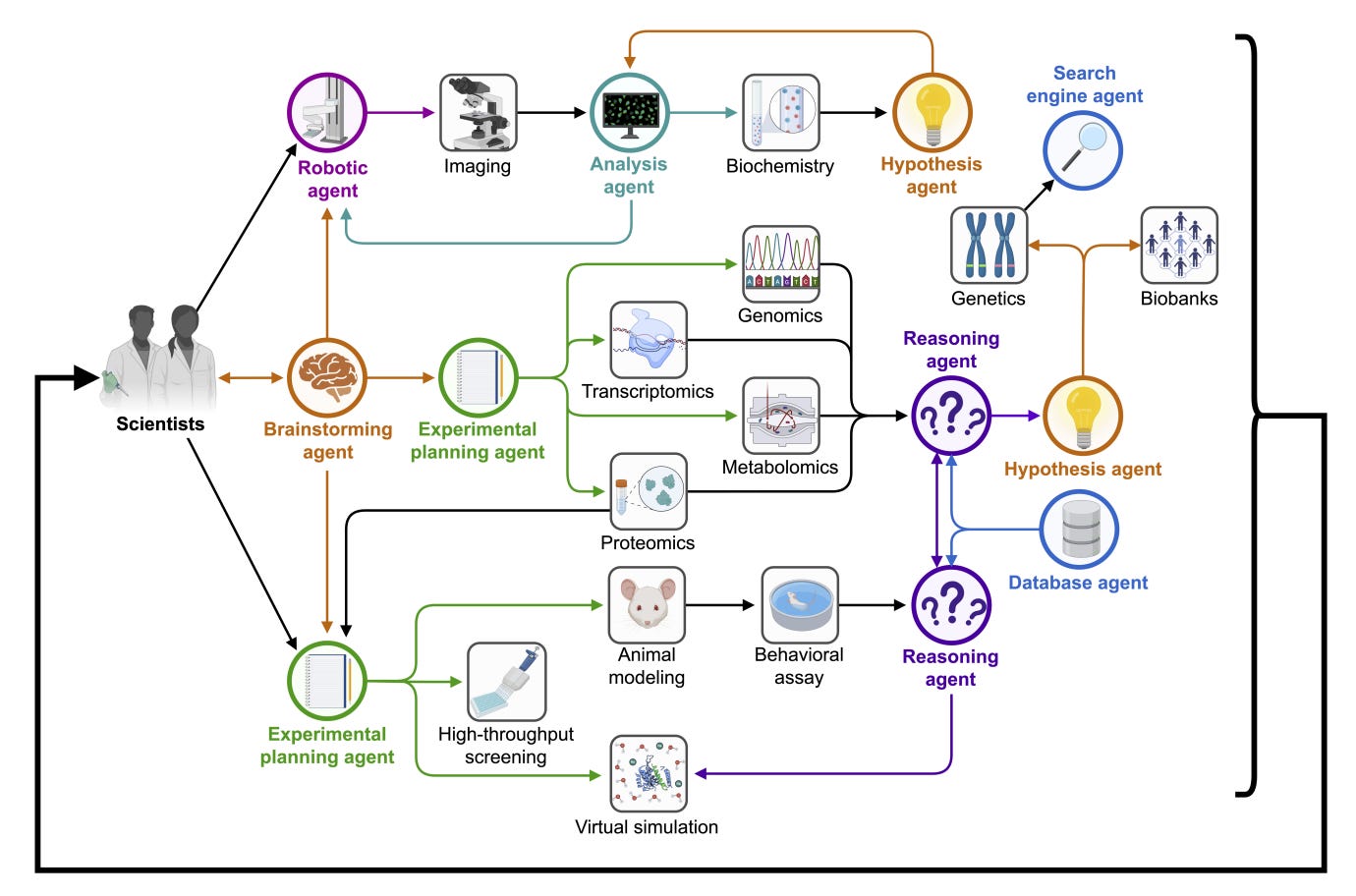
Empowering Biomedical Discovery with AI Agents [Gao et al., Zitnik Lab, arXiv, April 2024]
Why It Matters: The development of AI agents as outlined in the document is crucial for several reasons. First, these agents can significantly accelerate the pace of scientific discovery by automating complex, time-consuming processes that currently limit the efficiency of research workflows. Second, by integrating advanced AI tools with human intelligence, these systems have the potential to reach insights and innovations that might be impossible through human efforts alone. This could lead to breakthroughs in understanding and treating diseases, ultimately contributing to enhanced health outcomes worldwide. Furthermore, the responsible development and deployment of these AI agents also prompt an important dialogue on ethical AI use, ensuring that advancements in AI technology translate into tangible benefits for society without compromising safety or integrity.
The paper discusses the potential of "AI scientists" which are AI systems equipped with advanced machine learning tools and experimental platforms to drive biomedical research. These AI agents are designed to enhance the research process by integrating AI's capabilities to manage large datasets, explore hypothesis spaces, and perform repetitive tasks, while retaining human creativity and expertise. The agents employ large language models and generative models for structured memory and continual learning, aiming at applications ranging from hybrid cell simulation to the design of cellular circuits and development of new therapies.
Concept and Ambition: The envisioned AI scientists are sophisticated AI agents capable of undertaking comprehensive research tasks autonomously. They are not designed to replace human researchers but to collaborate intensively with them, enhancing creativity and efficiency. The aim is for these AI agents to someday achieve discoveries that could be worthy of Nobel Prizes, a challenge dubbed the "Nobel Turing Challenge."
Capabilities of AI Agents: These agents are equipped with advanced machine learning models, including LLMs and generative models, that enable them to perform tasks such as hypothesis generation, experimental design, and data interpretation. They possess structured memory for continual learning, meaning they can evolve their knowledge base without the typical AI problem of catastrophic forgetting. This is crucial for tasks that require adaptation to new data or changing environments.
Operational Efficiency: AI agents can streamline biomedical research workflows by automating the decomposition of complex scientific questions into simpler, manageable tasks. This capability allows them to handle large datasets efficiently and execute repetitive tasks that would be tedious and time-consuming for human researchers.
Applications: The applications of scientific AI agents are broad and impactful, ranging from simulating hybrid cells and controlling phenotypes programmatically to designing cellular circuits and developing new therapeutic approaches. These agents have the potential to revolutionize areas like drug discovery by predicting the effects of new compounds and optimizing therapeutic strategies based on vast amounts of genomic and biophysical data.
Ethical and Practical Considerations: The document also addresses the need for ethical guidelines and safety protocols in deploying AI agents. Given the profound capabilities of these agents, it is crucial to implement safeguards that prevent misuse and ensure that their operations remain under human oversight. Additionally, the potential for AI agents to inadvertently create dependencies where humans defer excessively to AI decision-making is highlighted as a risk that must be managed.
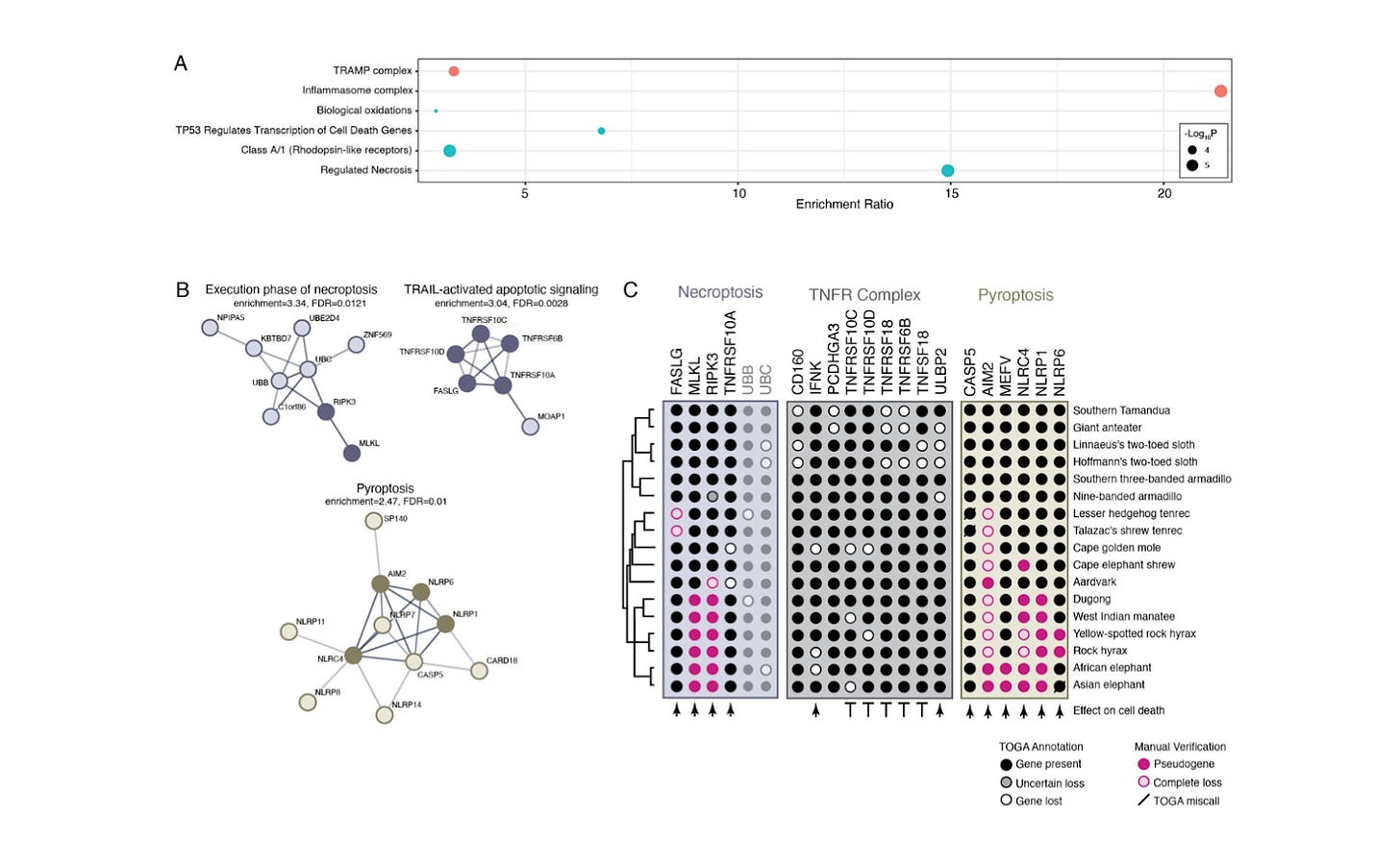
Pervasive loss of regulated necrotic cell death genes in elephants, hyraxes, and sea cows [BioRxiv, Birkmeier et al., 2024]
Why it matters: Certain animals, including elephants, have significantly enhanced cancer resistance. Previous work has elucidated that such animals have several extra tumor suppressors, including p53, but the dynamics of oncogenes has remained poorly resolved. In this paper, the authors demonstrate that Paenungulates have not lost oncogenes; rather, mechanisms regulating necrosis and pyroptosis (inflammatory cell death) are lost or derived, which may drive continued evolution of large bodies, cancer resistance, and susceptibility to microbial infection. This work sheds light on basic evolutionary biology and mechanisms leading to cancer resistance.
Paenungulates, including elephants and sea cows, are notable for their extremely large bodies and exceptional cancer resistance. Prior work has shown that elephants have unique anti-cancer cellular phenotypes that, as compared to smaller animals, 1) induce apoptosis at much lower levels of DNA damage, 2) are more resistant to oxidative stress, 3) have faster DNA damage repair rates, 4) are resistant to laboratory immortalization, and 5) have a seeming genetic barrier to inducing pluripotency. These traits have largely been explained by the presence of increased tumor suppressors, but the role of oncogenes has hitherto been unknown.
Here, the authors probed the genomes of 17 Afrotherian and Xenarthrian species to identify lost genes, and used a technique known as Dollo parsimony to reconstruct evolutionary trees. They found that – rather than losing oncogenes – these animals have lost necroptosis-mediating genes such as MLKL and RIPK3, alongside sensors that induce pyroptosis, such as IM2, MEFV, NLRC4, NLRP1, and NLRP6. What is remarkable is that these genes are heavily implicated in antimicrobial resistance; however, these animals do not appear to be overtly susceptible to microbial infection. This suggests that they have evolved alternative ways of activating pyroptosis (and other neutrophil-mediated killing mechanisms) in response to microbes. Thus this work suggests that loss of certain inflammasome genes may be tolerated, and can actually confer unexpected benefits, including cancer resistance.
Notable Deals
Nvelop launches with $100M to tackle delivery in gene editing medicines - the space has seen an explosion of tools such as CRISPR, base editing and prime editing but has been held back by effective methods of packaging and selectively delivering the therapies to the right targets in the body. The technology behind Nvelop is based on VLPs (virus-like particles) and the company is backed by the likes of Atlas, GV, 5AM, ARCH and F-Prime.
Diagonal Therapeutics launches with $128M to create antibodies that activate, rather than block, their targets. This may enable the activation of disabled cellular pathways that are contributing to disease progression. The biotech is seeded by Altas, Lightspeed and supporters include BVF and RA Capital.
Continuum prices IPO, raising $110M for neuroinflammatory drugs - the company has two drugs in testing: a wholly owned LPA1R antagonist for IPF and PPMS and an M1R antagonist partnered with J&J for RRMS and depression.
Seaport Therapeutics announces $100M for neuropsychiatric disorders - the biotech is the latest PureTech health creation which recently saw outsized success with the BMS buyout of Karuna.
Merck is acquiring Abceutis for $208M to make ADCs safer by using payload-binding selectivity enhancers that reduce toxicity to off-targets.
What we listened to
What we liked on socials channels
Events
Bio-IT World Conference & Expo | April 15-17, 2024 | Boston, MA & Virtual
Field Trip
Did we miss anything? Would you like to contribute to Decoding Bio by writing a guest post? Drop us a note here or chat with us on Twitter: @ameekapadia @ketanyerneni @morgancheatham @pablolubroth @patricksmalone











DecodingBio is one of the best things I have ever signed up for. So much interesting knowledge, presented in a very engaging and digestible way. Can’t get it enough of it! Thanks for your efforts :)