BioByte 007
AI-discovered ALS drug dosed in humans, all things fertility, advances to MRI, predicting drug responses in vitro, bacteria's role in autoimmune disease, and more
Welcome to Decoding TechBio, a writing collective focused on the latest scientific advancements, news, and people building at the intersection of tech x bio. If you’d like to connect or collaborate, please shoot us a note here. Happy decoding!
Happy November, y'all! Hope everyone had a great Halloween and is ready to transition from spooky to cozy. To recap, this week Verge Genomics announced it dosed the first patient with its AI-discovered ALS drug. This is a win for AI-drug discovery given the PIKFYVE inhibitor took four years to design and is among the first AI-discovered compounds in the clinic. We’ll keep our fingers crossed for the readouts. To keep it real, we also take a look at a paper investigating the similarities between AI-discovered drugs and existing FDA-approved therapies.
Before we get rolling, a huge thank you to everyone who has been sending us papers and posts they found interesting. Though we can barely keep up, we love hearing from you!
What we read
Blogs
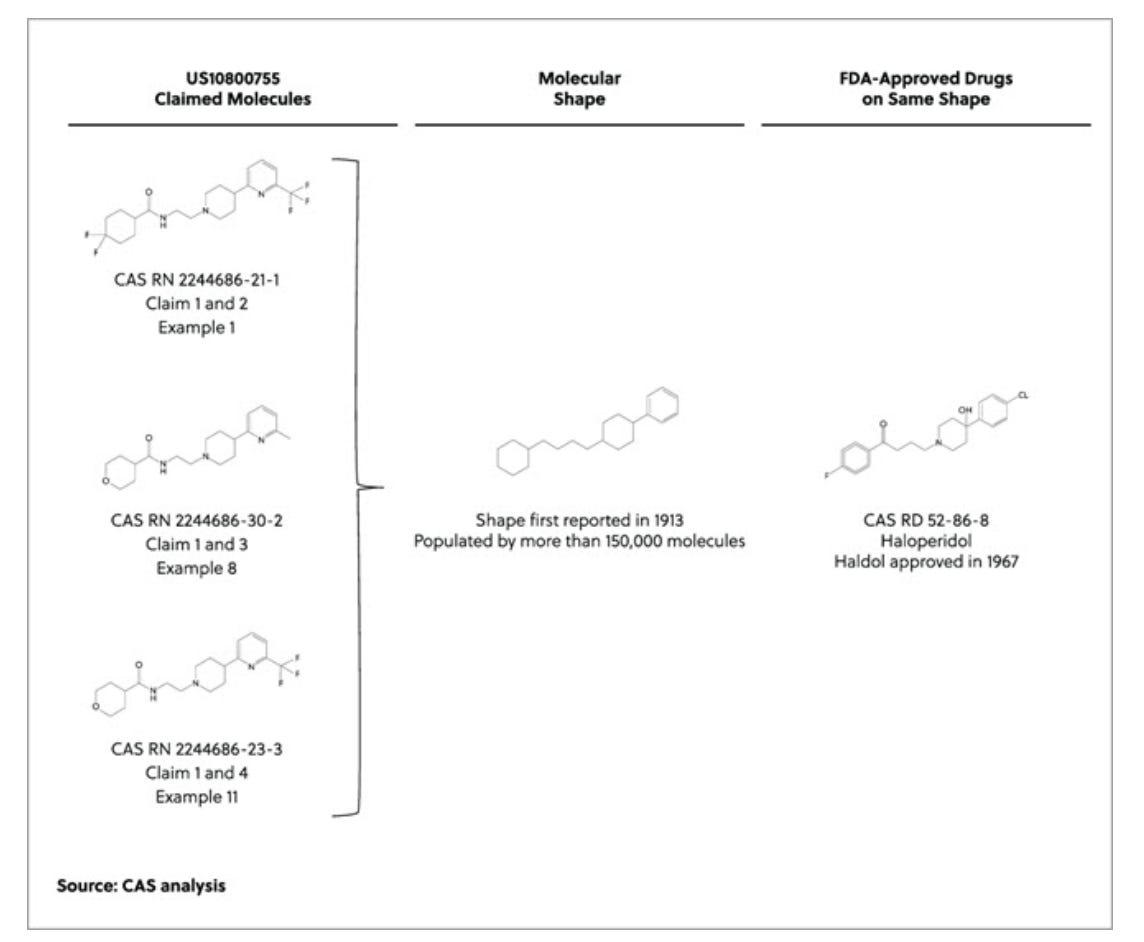
Assessing structural novelty of the first AI-designed drug candidates to go into human clinical trials [Todd Willis, Chemical Abstracts Service, 2021]
Why it matters: A commonly advertised benefit of AI-driven drug discovery is that AI enables the search of novel chemical space to discover entirely new chemical structures. A quantitative analysis by Todd Willis at CAS throws some cold water on this claim, and shows that the structure of a few clinical-stage AI-discovered drugs are similar to existing FDA-approved therapies. A thought-provoking analysis from last year that tested the novelty of AI-designed chemical structures for the first drug candidates to hit clinical trials. One hypothesis of AI-driven drug discovery is that AI enables the search of new chemical space and the discovery of new structures. To test this hypothesis, a quantitative method for assessing the uniqueness of the chemical structure was used to evaluate the structural novelty of three of Exscientia’s AI-designed drug candidates. The authors found that the chemical structures were pretty similar to existing FDA-approved Íor previously discovered drugs. So, while these molecules may have advantages relative to previously known chemical structures, the idea that AI in these scenarios had discovered entirely new chemistry doesn’t hold up. For additional context, see Derek Lowe’s blog on the topic.
Structural analysis of molecules in Exisentia’s 5-HT1a receptor agonist being investigated as a treatment for OCD. Note the structural similarity to Haloperidol, a first-generation antipsychotic approved by the FDA in 1967.
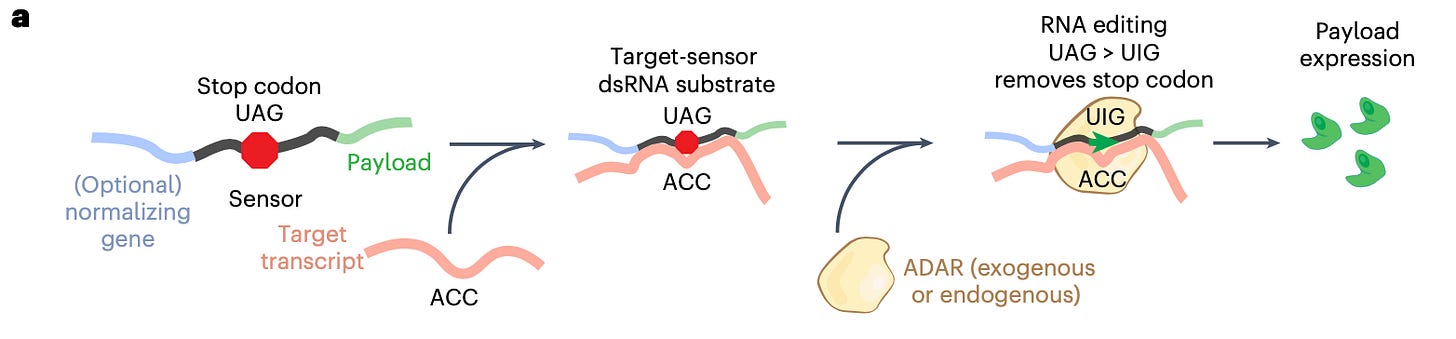
Broad and MIT scientists repurpose hot RNA editing tech as ‘radar’ with uses in cancer and brain research [Lei Lei Wu, Endpoints, 2022]
RNA-editing is capable of modifying disease states without permanently changing the genome. The ADAR system is the core RNA-editing platform for many biotechs including Shape Therapeutics, Wave Life Sciences, Korro Bio, and ADARx Pharmaceuticals.
In the past month, we have seen several papers cover novel RNA-sensing ADARs (see a more in-depth description here). Last week, the lab of Fei Chen of the Broad and joint lab of Jon Gootenberg and Omar Abudayyeh at MIT published their reprogrammable ADAR (RADAR) proof-of-concept paper:
“ [...] they showed that the technology could distinguish liver cells, and when attached to a cell death enzyme, selectively kill them. Importantly, RADARS could act as an RNA measuring tool without disrupting transcription or translation or cause significant off-target editing.”
A Plea for Making Virus Research Safer [Dr. Jesse Bloom, NYT Opinion 2022]
Dr. Jesse Bloom, a virologist at the Fred Hutchinson Cancer Center in Seattle, explores the “double-edged” sword of modern virology as it relates to our safety. As researchers explore and manipulate potential pandemic viruses, adding “gain of function” mutations that could make candidates more transmissible, Dr. Bloom heeds caution that the risk-benefit tradeoff of such methods may not justify continued pursuit.
The article links to a powerful memo from the Center for Health Security: “Recommendations to Strengthen the US Government’s Enhanced Potential Pandemic Pathogen Framework and Dual Use Research of Concern Policies.”
Biotech begins human trials with drug discovered using AI [Jamie Smyth, FT, 2022]
Verge Genomics has dosed its first patient with a novel therapy named VRG50635 to target ALS. Last year, Verge raised a ~$100M Series B led by BlackRock. It also signed a deal with Eli Lilly with an upfront payment of $25M and potential milestone and royalty payments worth $694M.
Verge has built a disease map, across the ‘omics stack, based on diseased human brain tissue to discover the underlying causative mechanisms of ALS and other neurological diseases. The company states that the loss of endolysosomal function is a putative mechanism that causes ALS and that drugging their proposed target (PIKFYVE) should ameliorate the condition.
Alice Zhang describes to BioPharma Dive how the company leverages AI to find novel targets:
“What we did differently with this program is instead of just exploring the solution through trial and error, we actually collected large repositories of human tissues. We’ve collected — not just for ALS — over 8,000 human samples. These are brains and spinal cords where we’ve sequenced each sample all the way from DNA to RNA to protein, and paired that with clinical data.
Then we develop machine learning tools to mine this data and create a comprehensive map of human biology. We use it to identify new intervention points to target using drugs that we develop internally in our pipeline”.
Fertility White Paper [Willy Chertman, 2022]
This white paper is possibly the most comprehensive resource we’ve seen on all things fertility—covering everything from declining rates to reproductive aging to in vitro gametogenesis. Willy also includes notes from his interview with experts and leaders as well as a list of fundable projects in the area. If you have any interest in fertility and reproductive health, this is the white paper for you. And if you are short on time, here’s the Twitter thread version :)
Academic Papers
In vivo direct imaging of neuronal activity at high temporospatial resolution [Toi et al., Science, 2022]
All neuroimaging techniques are trade-offs between spatial and temporal resolution. EEG is able to resolve neuronal activity with millisecond precision, but has poor spatial resolution. Functional MRI can localize neuronal activity (indirectly, via blood flow) with higher spatial resolution on the order of millimeters, but because fMRI measures the blood-oxygen-level-dependent (BOLD) signal, which is based on blood flow that lags neuronal activity, the temporal resolution is slower on the order of seconds. A major advance published this month in Science describes a technique for high temporal measurement of neuronal activity in rats using a 9.4 tesla MRI. The method works by hacking an MRI machine to acquire partial images at short time intervals rather than the longer, full-brain images typically acquired in fMRI. The team verified using optogenetics and electrophysiology that they were in fact measuring neuronal activity. The authors argue that they are not picking up BOLD signals, but rather changes in membrane potential of neurons, potentially via changes in water concentration on the membrane’s surface. Additional studies are needed to fully understand the mechanism, but this is a huge advance for the field of non-invasive neuroimaging.
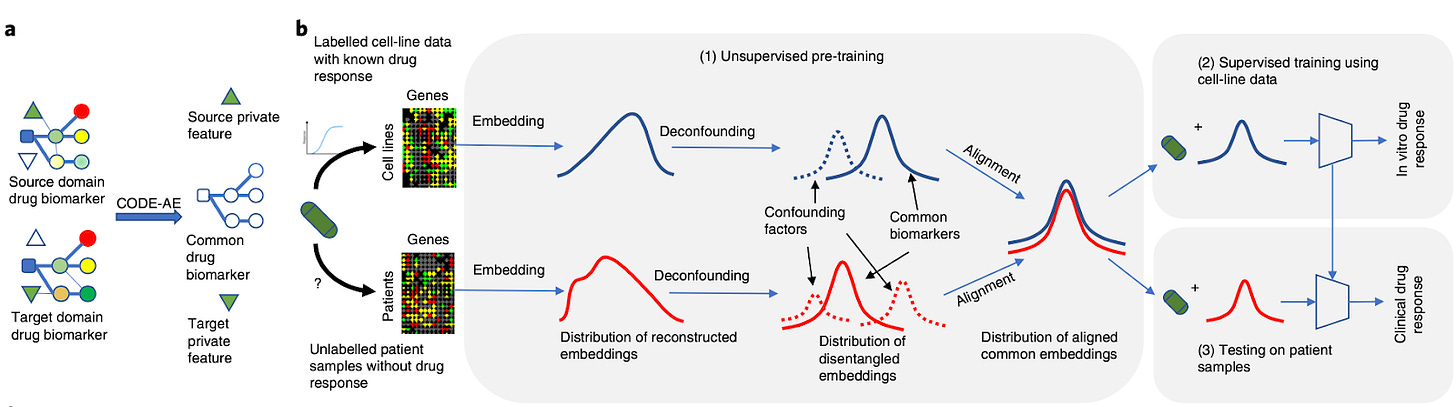
A context-aware deconfounding autoencoder for robust prediction of personalized clinical drug response from cell-line compound screening [He et al., Nature Machine Intelligence, 2022]
Why it matters: Predicting drug response in humans based on molecular biomarkers has already been extremely impactful by stratifying patient populations and thus improving response rates (see HER2 and Traustuzumab for breast cancer). However, for more complex mechanisms of action, it has been problematic to predict human response based on cell-line compound screening data. This is due to context-specific patterns and confounding factors. In this paper, the authors implemented a new model that can take into account the ‘context’ of data sets to significantly improve the accuracy of the in vitro to human drug response predictions.The authors have developed a context-aware deconfounding autoencoder (CODE-AE) that can extract intrinsic biological signals masked by context-specific patterns and confounding factors. The underlying assumption of existing machine learning methods is that the data distribution of training data and unseen testing data are the same. In the context of drug response prediction from in vitro screens, this means that when a different set of data is used on an ML algorithm trained on a data set with a different context, it is unlikely to predict accurately (named the out-of-distribution [OOD] problem). The authors state that “extensive benchmark studies demonstrate the advantage of CODE-AE over the state of the arts in terms of both accuracy and robustness. When CODE-AE is applied to predict drug responses for patients in The Cancer Genome Atlas, the predictions are largely consistent with existing clinical observations.”
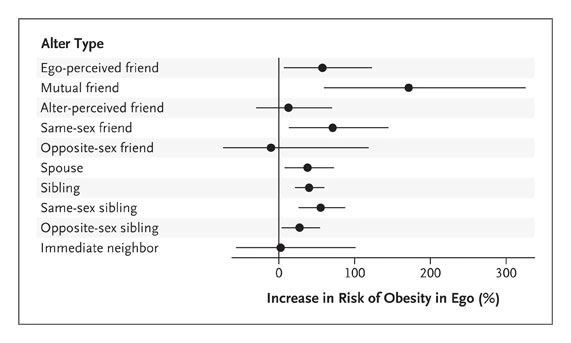
The Spread of Obesity in a Large Social Network over 32 Years [Christakis, et al., NEJM, 2007]
This paper, though fifteen years old, was resurfaced by a friend during a conversation about lifestyle affects on medicine and how we can better address lifestyle-interventions as therapeutics. Perhaps it is no longer a shocking conclusion, but the extent to which social networks affect the spread of obesity is noteworthy. The longitudinal study concludes that a person’s chance of becoming obese increased by 57% if he/she had a friend who became obese in a given period of time. Similar results followed for other social relationships including spouses and siblings. Interestingly, the results did not hold true for neighbors (based on geography), which underscores the value of close social relationships in this spread. There are many factors that play into the spread of obesity as a social network including cultural tolerance, social influence on activity and habits, and even potential physiological imitation (read: brain stimulation).
Clonal IgA and IgG autoantibodies from individuals at risk for rheumatoid arthritis identify an arthritogenic strain of Subdoligranulum [Chriswell et al, Science Translational Medicine, 2022]
A new paper out of the University of Colorado has found a possible link between rheumatoid arthritis and cross-reactivity to a few strains of bacteria commonly found in the gut.
Why it matters: There is little known about the root cause of most autoimmune disorders including rheumatoid arthritis. While these diseases most commonly affect women and there are often certain "kickstarter" events such as infection, there is still much mystery on how autoantibodies are developed and to which antigens. This paper sheds some light on a possible link between RA and bacterial cross-reactivity. The authors studied the blood of rheumatic patients to understand what makes certain groups of people develop autoantibodies associated with the autoimmune disease. They found that strains of the Subdoligranulum bacteria reacted significantly with monoclonal antibodies from the blood samples. The authors also conducted mouse studies where they inoculated mice with the bacteria and tracked the development of RA phenotype including T-cell expansion. The results of the study hint that bacterial strains, often found in the gut, may be involved in triggering rheumatoid arthritis and likely other autoimmune disorders. What is still unknown is how these bacteria interact to switch the immune system into hyperdrive and what other strains of bacteria (and potentially other organisms?) may play a role as well.
What we listened to
Bob Langer shares his perspectives on applications of artificial intelligence in biotechnology, emphasizing the importance of understanding the biology. He also shares perspectives on the current microenvironment and what technologies he’s most excited about for the years ahead.
Michael Levin talks about developmental biology, evolution, and more with Lex Friedman.
Notable Deals
Alto Neuroscience Announces $35 Million Series B Financing. Alto Neuro is a precision neuropsychiatric platform that uses a multi-modal (EEG, behavioral measures, wearables, genetics) biomarker platform to develop patient-targeted therapeutics. Spun out of Amit Etkin’s lab at Stanford, and built upon a body of landmark publications, such as the identification of therapeutically-relevant subtypes of psychiatric disorders using resting state EEG and the use of EEG to predict antidepressant response in major depression. The company raised a $35m Series B led by Lightswitch Capital and partners of Alkeon Capital, with participation from other new investors including Sobrato Capital, Novartis Pharma AG, Valor Equity Partners, Korify Capital, Vine Ventures, and Gaingels. The financing also included participation from existing investors Apeiron Group, WhatIf Ventures, Windham Venture Partners, and others.
What we liked on Twitter
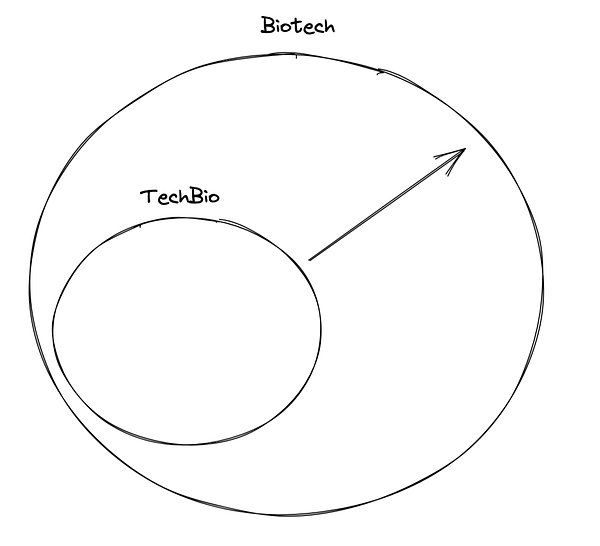
Lot’s of Twitter debate on what TechBio encompasses this week. While we tend to agree with Elliot’s diagram, here are some highlights from the discussion.



And in other news:




Events
Founder Led Bio Summit | Oct 31-Nov 3 | Virtual, SF, Boston
Pillar/Petri are back with another amazing Founder Led Bio Summit. Tune in to hear from your favorite bio leaders and attend the happy hours in SF and Boston if you’re around. More details here.
TechBio Cocktails | Nov 9 | NYC
A few of us are organizing a happy hour for founders, operators, scientists, and investors in NYC on November 9. If you happen to be in the area, RSVP here.
Field Trip

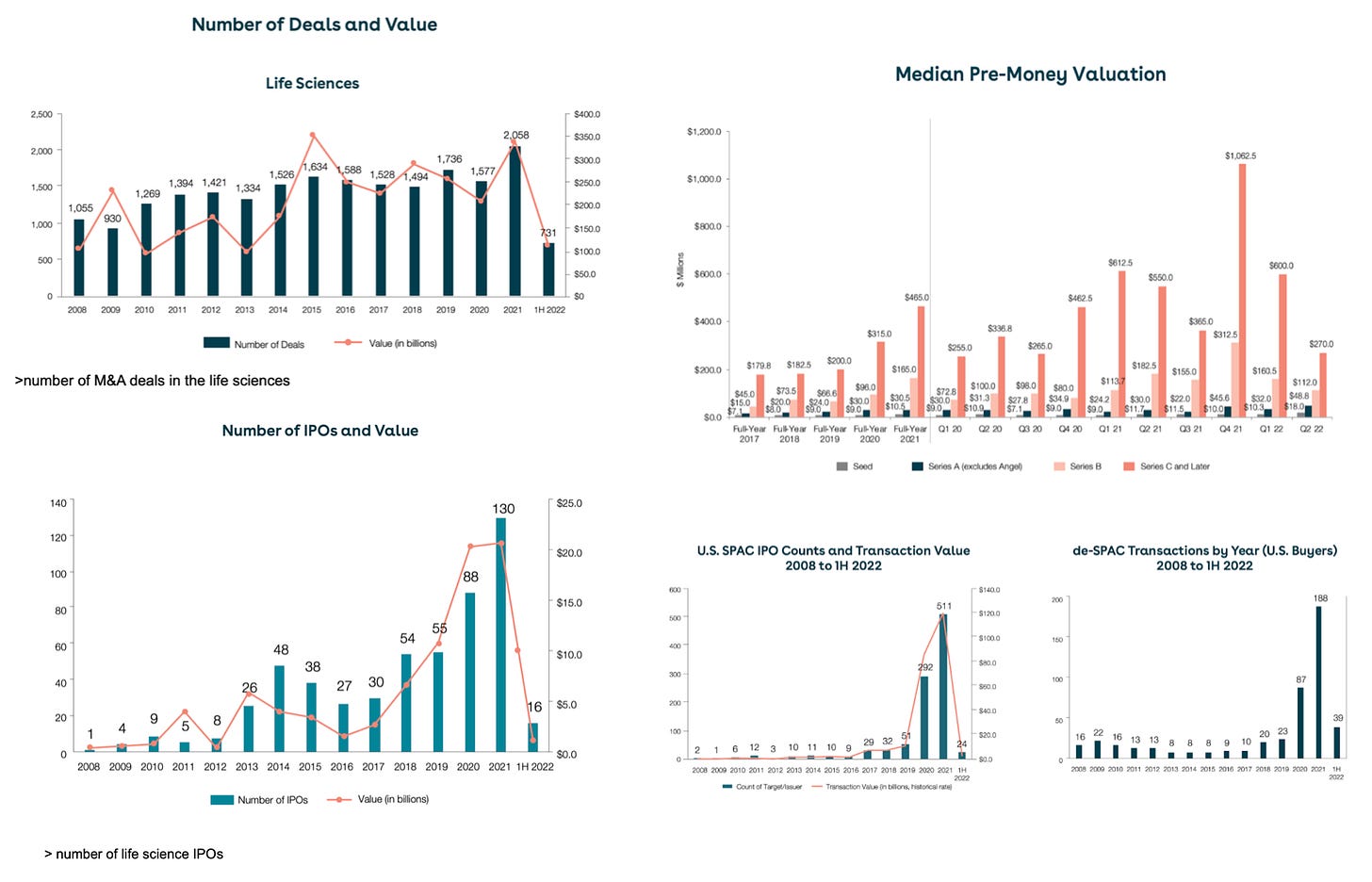
Wilson Sonsini’s 2022 Mid-Year Technology and Life Sciences "Non" IPO Report in 5 charts:
Did we miss anything? Would you like to contribute to Decoding TechBio by writing a guest post? Drop us a note here or chat with us on Twitter: @ameekapadia @pablolubroth @patricksmalone @morgancheatham @ketanyerneni