BioByte 071: Unanswered questions, paused base-editing therapy, challenges with ASOs, GPT-4 for cell type annotation, the scientific journey behind Ozempic
Welcome to Decoding Bio, a writing collective focused on the latest scientific advancements, news, and people building at the intersection of tech x bio. Happy decoding!
What we read
Blogs
Some questions about biotech that I find interesting [Alex Telford, March 2024]
Must-read biotech writer and observer Alex Telford is back with a list of biotech-themed questions. Some highlights:
Has the outsourcing of pharma R&D been a net positive or negative for the industry? Most new drugs are now discovered by small biotechs, not large pharma, and CROs and CDMOs run much of the development and manufacturing. What data exists that this is a good thing for the industry?
The decreasing cost curve of sequencing the human genome is well known, but has the cost of manufacturing advanced biologics such as cell and gene therapies and mRNA similarly decreased? Does biomanufacturing obey Wright’s law (the cost of producing technology decreases as a fixed percentage of increases in cumulative production volume) like solar, computers, and batteries?
What is the right spatial scale for analysis for drug discovery? Most of drug discovery focuses on the smaller scale at the level of proteins, but lots of disease-relevant biology happens at more meso- or macro- spatial scales such as biological networks or tissues. I would add that there is also a temporal dimension and scale that is mostly ignored (eg how protein interactions and dynamics unfold over time), that is mostly ignored in drug discovery.
The biotech field is aware of all of the false positives of preclinical testing; drugs that treat disease in animal models, but fail in clinical trials. But less discussed is the false negative rate - what’s the best drug that never got developed because it was deprioritized based on preclinical data? This is an insidious problem: the absence of evidence (a drug failing preclinical testing) is not necessarily evidence for absence (the drug not working in humans).
Verve pauses base editing study after treatment side effect [Ned Pagliarulo, Biopharma Dive]
In mid-2022, Verve’s PCSK9 therapy became the first base editing therapy to ever enter the clinic. On Tuesday this week, Verve disclosed that the company was pausing enrollment after a participant developed lab test abnormalities for its VERVE-102 programme.
The study participant was asymptomatic from the lab test abnormalities which could point to potential liver damage and thrombocytopenia. After 48h, and a course of oral steroids, the patient fully recovered. Verve believes the LNP system is what caused the abnormalities. Verve also updated efficacy data for VERVE-101 which has shown 46% reduction of LDL-C amongst the first five participants (treated with a 0.45 milligram per kilogram of body weight dose).
End of the Beginning [Eric Minikel, CureFFI.org]
It has been three months since Ionis began recruiting for their ASO trial in prion disease; since then, nine trial sites have been recruited, and really marks a significant milestone in the treatment of this disease. Prions are relatively new, as they were only discovered in the early 1980s by Stanley Prusiner, and were a therapeutic graveyard.
Decades of work had resulted in failures, given the resistance and stability of these pathogenic molecules - no matter which modality or mechanism you threw at prions, they failed to modulate disease progression.
Ionis’s drug finally fills in the missing pieces of drugs prior, by targeting the root cause of disease via lowering the amount of the PrP molecule in the brain. This approach appears to work in animal models; importantly, we are able to measure PrP in CSF and tissue of preclinical models and humans, meaning we have the ability to truly understand PK/PD considerations. Although continuing to translate this from bench to bedside will take Herculean efforts, this trial is a testament to how far we have progressed in our understanding of this incurable disease. Although the field is cautiously optimistic, failure is the de facto outcome: Eric goes on to explain many potential reasons why the Ionis trail could fail and touches upon the challenges that plague ASOs and will require further evaluation, such as biodistribution, potency, trial design, and more.
How to make an old immune system young again [Ledford, Nature News, March 2024]
In a pioneering study published in Nature, scientists have rejuvenated the immune systems of aged mice by targeting and reducing pro-inflammatory stem cells with antibodies. This intervention not only bolstered the mice's defenses against viral infections and diminished signs of inflammation but also hinted at a more youthful immune functionality. The research, spearheaded by Irv Weissman’s team at Stanford University, marks a crucial advancement in understanding how aging affects blood stem cell populations and their roles in immune responses. Although human applications may be years away, the similarities in stem-cell biology between mice and humans suggest promising potential for aging and immune system research. This work opens exciting avenues for future exploration, including the potential for human trials within the next three to five years, offering a glimpse into new strategies to combat the decline in immune function with age.
How scientists are making the most of Reddit [Doctor-Loeb, Nature, March 2024]
Nearly 18 months after Elon Musk's acquisition of Twitter, now renamed X, the platform has seen a 15% drop in daily active users and a drastic 80% workforce reduction. In response, scientists are migrating from X to alternative social media platforms, with many finding a new community on Reddit, a platform established in 2005 that predates Twitter. Reddit boasts 73 million daily active users across over 100,000 subreddits, facilitating discussions on a wide array of topics, including science-focused communities. The site's structure allows for pseudoanonymous interaction through upvoting, downvoting, and commenting, promoting a diverse exchange of ideas and information. Despite recent challenges in data accessibility, Reddit remains a valuable resource for non-commercial researchers and academics, offering a blend of networking, research collaboration, and public engagement opportunities. Scientists like Yvette Cendes and students like Kevin Ortiz Ceballos highlight the platform's capacity for impactful science communication and community building, underscoring its growing relevance in the shifting social media landscape.
US biosecurity starts at home, with insurance reform aimed at making innovation affordable [Kolchinsk and Cameron, Rapport Bio, March 2024]
Congress is increasingly concerned about the United States' dependency on China's biotechnology industry, particularly the risk of the Chinese Communist Party accessing Americans' genetic data and US taxpayer funds supporting CCP-affiliated companies. Efforts are underway to disconnect federally funded projects from Chinese entities considered problematic, including major firms like BGI and WuXi AppTech. This initiative is part of a broader strategy to safeguard and enhance the United States' leading position in biomanufacturing and biotechnology, as underscored by the Biotechnology Innovation Organization's (BIO) support for the BIOSECURE Act. BIO's stance, especially after initially appearing to back companies like WuXi, signifies a pivotal shift towards prioritizing national security and biotech innovation within the US, highlighting the strategic importance of domestic biomedical research and development.
However, the BIOSECURE Act alone may not suffice to ensure the US remains at the forefront of biomedical innovation. The debate extends into the realm of drug affordability and the impact of government-imposed price controls on innovation. The underlying concern is that without affordable access to treatments, public support for biomedical innovation could wane, irrespective of the country of origin. This sentiment suggests a need for policies that not only foster innovation but also ensure treatments are accessible and affordable for Americans. The US's current reliance on foreign firms for critical R&D services and supplies further complicates the situation, raising questions about the resilience of the US biotech industry to geopolitical tensions and underscoring the necessity for a comprehensive strategy that includes insurance reform, supply chain diversification, and incentives for domestic innovation to secure the biomedical future of the US against emerging global competitors.
Academic papers
Assessing GPT-4 for cell-type annotation in single-cell RNA-seq analysis [Hou et al., Nature Methods, March 2024]
Why it Matters: The ability of GPT-4 to automate cell type annotations with high accuracy and concordance with manual methods represents a significant leap forward in the field of genomics and transcriptomics. This advancement touches on:
Efficiency and Accessibility: Automating the labor-intensive and expertise-demanding task of cell type annotation not only saves time but also makes cutting-edge genomics research more accessible to a broader range of scientists, including those who may lack specialized training in bioinformatics.
Enhanced Accuracy and Reproducibility: By leveraging the computational power of GPT-4, researchers can achieve consistent and reproducible annotations. This consistency is crucial for validating experimental results and advancing our understanding of complex biological systems.
Interdisciplinary Integration: The integration of advanced AI models like GPT-4 into genomics signifies the growing intersection of computer science and biology. This interdisciplinary approach is paving the way for novel discoveries and innovations in medical research, drug development, and beyond.
Scalability for Future Challenges: As the volume of genetic data continues to grow exponentially, traditional manual annotation methods will become increasingly untenable. The scalable nature of GPT-4's automated annotations ensures that the scientific community can keep pace with this data deluge, unlocking new insights into disease mechanisms, genetic disorders, and therapeutic targets.Researchers Wenpin Hou and Zhicheng Ji have demonstrated that GPT-4 can significantly streamline cell type annotation in single-cell RNA sequencing (scRNA-seq) analysis by accurately utilizing marker gene information. Their findings, backed by an R software package named GPTCelltype, showcase GPT-4's strong concordance with manual annotations across a wide range of tissue and cell types, signaling a shift towards semi- or fully automated annotation processes. This advance could dramatically reduce the manual effort and expertise traditionally required, offering a cost-efficient and easily integrable solution into existing scRNA-seq analysis pipelines like Seurat. Moreover, GPT-4's broad training on diverse data sets and its interactive nature for user-guided refinement further underline its potential to improve annotation accuracy and efficiency across various biomedical research contexts.
Depleting myeloid-biased haematopoietic stem cells rejuvenates aged immunity [Nature, March 2024]
Why it matters: Given the conservation between mice and humans of HSCs, this early study could support the development of therapies to rejuvenate the blood-forming system in patients, strength adaptive immunity and reduce inflammation.Inflammation and susceptibility to infection are two key dangers in ageing animals. A team at Stanford led by Irv Weissman has found that treating aged mice with antibodies against a certain class of stem cells strengthened the old rodents’ responses to viral infection and lowered signs of inflammation. This class of stem cell (my-HSCs) are biased towards the production of myeloid cells and their predominance in the haematopoietic stem cell class increases with age. This decreases lymphopoiesis which is required for healthy adaptive immune responses and increases myelopoiesis which contributes to a pro-inflammatory phenotype. Thus, the team sought to restore the balance of my-HSCs in aged mice by antibody-mediated depletion. The results showed increased lymphocyte progenitors and naive cells, decreased markers of lymphocyte exhaustion and decreased inflammatory mediators. Importantly, HSC rebalancing also improved protective immunity in aged mice to infection.
Human lung cancer harbors spatially organized stem-immunity hubs associated with response to immunotherapy [Nature, March 2024]
Why it matters: PD-1 therapies have been life-changing for millions of patients, however many treated with such inhibitors do not respond and researchers have been trying to understand why. An improved ability to stratify patients based on responders and non-responders will dramatically decrease needless spending, improve clinical trial outcomes and reduce patient suffering from treatments that have positive effect. Indeed, a poor ability to stratify patients based on response to immunotherapy is one of the major hurdles holding this class of therapeutics back.Scientists at the Broad Institute have been studying immune cells in tumors from 68 people with NSCLC prior to PD-1 inhibitor treatment. The team mapped the cell’s genetic activity and the location of the cells within the tumors using multiplex RNA-based and protein-based imaging, as well as spatial transcriptomics. The team found that responders to PD-1 therapies tended to have a type of immunity hub called a stem-immunity hub enriched for stem-like TCF7+PD-1+CD8+ T cells, activated CCR7+LAMP3+ dendritic cells and CCL19+ fibroblasts as well as chemokines that organize these cells. A clear implication of this work is that the presence of an immunity hub can be used as a biomarker to predict response to immunotherapy. The current standard biomarker for prediction of immunotherapy response is PD-L1 antibody staining, but it can be inaccurate and difficult to use.
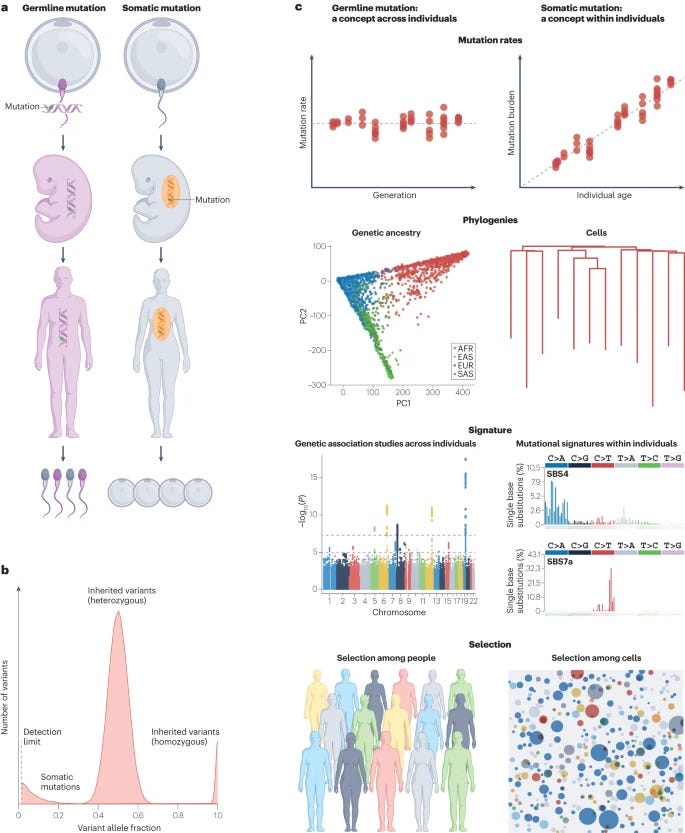
Genetic variation across and within individuals [Yu et al., Nature Reviews Genetics, March 2024]
Why It Matters: Understanding the dynamic interplay between germline and somatic genetic variations is crucial for several reasons. First, it enhances our comprehension of the genetic foundations of various diseases, including cancers and inherited disorders, offering potential pathways for new treatments and diagnostic approaches. The knowledge of how these variants contribute to disease mechanisms can lead to personalized medicine strategies, where treatments are tailored based on an individual's unique genetic makeup. Moreover, insights into somatic mutations across different tissues can reveal patterns of aging, tissue regeneration, and the development of age-related diseases, contributing to our understanding of human biology and longevity. Lastly, the advancements in detecting these genetic variations open avenues for preventive healthcare, allowing for the identification of disease predispositions early in life and the implementation of targeted interventions to mitigate these risks.The recent review article delves into the intricacies of genetic variation within and across individuals, highlighting the significant roles of germline variants and somatic mutations in human traits and disease risks. Germline variants, which are present from conception, differ between individuals and accumulate over generations. Somatic mutations, on the other hand, are acquired throughout an individual's life, creating a mosaic of variations within due to various intrinsic and extrinsic factors. Recent advancements in detection methods and the expansion of resources for association studies have broadened our understanding of these genetic variations, their mutational processes, and their interplay. The review underscores the differences in mutation rates, types, and patterns between germline and somatic variants, and it explores their combined impact on human health and disease, providing insights into inherited conditions, cancer development, and other non-oncologic diseases influenced by somatic mutations.
Notable Deals
Start-ups:
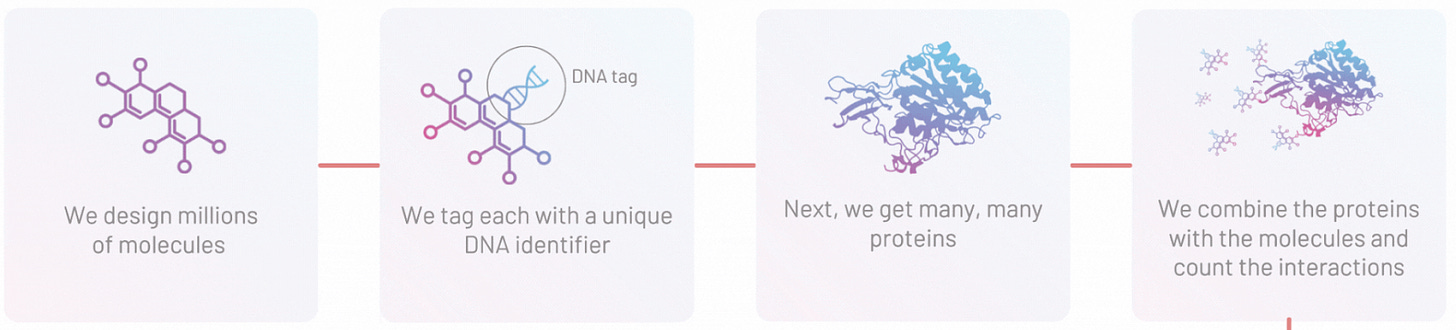
Leash Bio launches with a novel dataset and a novel business model - The ex-Recursion and Salt Lake City based biotech has been building a proprietary dataset of protein-molecule interactions designed for machine learning and believes firmly that lack of sufficient data has held back machine learning efforts in drug discovery. Leash is running an open-contest calling machine learning researchers to train their models on Leash’s dataset, setting a new bar for the industry and aiming to remove some of the opaqueness around performance.
Financings:
Zola Therapeutics emerges from stealth to tackle cancer immunotherapy with an unusual business model - the biotech is led by Art Krieg who co-founded and sold Checkmate to Regeneron. The idea came from previous work at Checkmate, and is based on packaging nucleic acids into lipid nanoparticles to stimulate T cell responses. The biotech will be funded by its founding team till preclinical testing and pharma deals will be used to progress the company beyond. This unusual model is based on a desire to retain full control over the journey of the biotech.
Alterome Therapeutics raises $132M series B to bring its two programmes (AKT1 and KRAS) to the clinic in the next year. The biotech has generated 75 proprietary protein-ligand crystal structures to give it in an edge in tackling these targets.
Onego bio raises $40M to drive commercialisation and manufacturing of egg protein via precision fermentation - Onego’s Bioalbumin is bioidentical to the major protein in chicken egg, ovalbulmin, and aims to replace this in industrial food preparations.
Metsera launches with $350M - whilst in stealth, the obesity biotech has in-licensed six obesity drug candidates, acquired a London-based biotech and is targeting 7 IND filings in the next two years.
Neurosterix emerges with $63M to advance allosteric modulators for neurological diseases
Obsidian Therapeutics raises $160M to push its TIL therapies further into clinical trials
Spatial AI biomarker startup Nucleai raises $14 million led by Merck’s VC arm
Partnerships:
Merck KGaA forms alliance with Caris Life Science to discover and design new ADCs with $1.4B in potential biobucks
Ipsen inks $900M deal with Sutro to pick up its first ADC asset
M&A:
Genmab acquires ProfoundBio for $1.8B - the first major acquisition for the Danish antibody giant aims to apply its expertise in bispecifics and antibody technologies towards the development of ADCs, which Profound has been developing since 2018. The Genmab team were impressed with Profound’s linker and payload technology as well as its assets which recently showed impressive clinical data. Genmab also foresees combinations between the two companies assets as they emerge into Phase III.
Ginkgo acquires Modulus Therapeutics - the rationale is to accumulate technologies that can help boost the safety and potency of cellular medicines, increasing Ginkgo’s capabilities in cell therapy. Modulus has a database of NK-specific and T-cell specific CAR libraries and switch receptor designs which can enable more precise control over activation and targeting.
What we listened to
In case you missed it
What we wished we knew entering the aging field [Age1 and Maggie Li]
What we liked on socials channels
Participate in Chrissy’s thought experiment here
Events
Counting Leash Bio’s new Kaggle competition as an event :) Check it out here if you want to predict some small molecule - protein interactions for a $50K prize!
Field Trip
Going down rabbit-holes with Suno AI
Did we miss anything? Would you like to contribute to Decoding Bio by writing a guest post? Drop us a note here or chat with us on Twitter: @ameekapadia @ketanyerneni @morgancheatham @pablolubroth @patricksmalone










Your 3rd thought is something I have been thinking about as well. Who and what will build dynamic systems models (PDEs, ODEs and SDEs) to attempt to simulate and possibly predict drug efficacy and toxicity. I have seen one company attempting this: Syntensor. Are there others?
Super interesting stuff!! Ty!!!